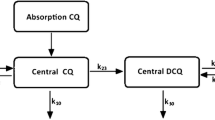
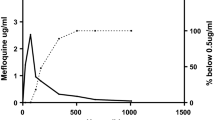
Summary
The pharmacokinetics of primaquine (PQ) and its major carboxylic acid metabolite (PQC) have been studied in seven Indian patients withP. vivax malaria following PQ 15 mg/day p.o. for 14 days. After a single oral dose on Day 1, a mean peak blood concentration of 50.7 ng/ml PQ was attained after 2.3 h, which declined monoexponentially with a half-life of 5.6 h. The mean total body clearance was 37.6 l/h and the volume of distribution was 2921. The mean renal excretion (0–24 h) of the drug was only 0.54% of the dose and renal clearance was 0.189 l/h. Following chronic administration, none of the pharmacokinetic parameters was affected, and a steady state blood concentration of 2.5–4.2 ng/ml PQ was attained. After the first dose of PQ, PQC had a mean area under the blood concentration — time curve 11-fold higher than that of the parent drug. In contrast to the rapid distribution and elimination of PQ, the metabolite showed a longer mean residence time and accumulation in the body. The mean Cmax and AUC of the metabolite on Day 14 were 48 and 40% higher than the corresponding Day 1 values. The metabolite could not be detected in urine at any time in any patient. PQ and its metabolite did not show any accumulation in blood cells.
Similar content being viewed by others
References
World Health Organisation Technical Report Series 711 (1984) Advances in malaria chemotherapy. World Health Organisation, Geneva
Rollo IM (1980) Drugs used in the chemotherapy of malaria. In: Gilman AG, Goodman LS, Gilman A (eds) Goodman and Gilman's — The pharmacological basis of therapeutics, 6th edn Macmillan, New York, p 1051
Ward SA, Edwards G, Orme ML'E, Breckenridge AM (1984) Determination of primaquine in biological fluids by reversed-phase high-performance liquid chromatography. J Chromatogr 305: 239–243
Bhatia SC, Revankar SN, Bharucha ED, Doshi KJ, Banavalikar MM, Desai ND, Subrahmanyam D (1985) Determination of the antimalarial primaquine in whole blood and urine by normal-phase high-performance liquid chromatography. Anal Lett 18 (B13): 1671–1685
Mihaly GW, Ward SA, Edward G, Nicholl DD, Orme ML'E, Breckenridge AM (1985) Pharmacokinetics of primaquine in man. I. Studies of the absolute bioavailability and effects of dose size. Br J Clin Pharmacol 19: 745–750
Ward SA, Mihaly GW, Edwards G, Looareesuwan S, Phillips RE, Chanthavanich P, Warrell DA, Orme ML'E, Breckenridge AM (1985) Pharmacokinetics of primaquine in man. II. Comparison of acute vs chronic dosage in Thai subjects. Br J Clin Pharmacol 19: 751–755
Mihaly GW, Ward SA, Edwards G, Orme ML'E, Breckenridge AM (1984) Pharmacokinetics of primaquine in man: Identification of the carboxylic acid derivative as a major plasma metabolite. Br J Clin Pharmacol 17: 441–446
Gibaldi M, Perrier D (1982) Noncompartmental analysis based on statistical moment theory. In: Pharmacokinetics, 2nd edn Marcel Dekker, New York, pp 409–416
Greaves J, Price Evans DA, Gilles HM, Fletcher KA, Bunnag D, Harinasuta T (1980) Plasma kinetics and urinary excretion of primaquine in man. Br J Clin Pharmacol 10: 399–405
Fletcher KA, Price Evans DA, Gilles HM, Greaves J, Bunnag D, Harinasuta T (1981) Studies on the pharmacokinetics of primaquine. Bull WHO 59: 407–412
Back DJ, Purba HS, Staiger C, Orme ML'E, Breckenridge AM (1983) Inhibition of drug metabolism by the antimalarial drugs chloroquine and primaquine in the rat. Biochem Pharmacol 32: 257–263
Back DJ, Purba HS, Park BK, Ward SA, Orme ML'E (1983) Effect of chloroquine and primaquine on antipyrine metabolism. Br J Clin Pharmacol 16: 497–502
Breckenridge A, Awadzi K, Back DJ, Edwards G, Gilles H, Orme ML'E, Ward S (1984) Drug metabolism and tropical disease. Biochem Soc Trans 12: 90–95
Author information
Authors and Affiliations
Additional information
Communication No.797 from Hindustan CIBA-GEIGY Limited, Research Centre
Rights and permissions
About this article
Cite this article
Bhatia, S.C., Saraph, Y.S., Revankar, S.N. et al. Pharmacokinetics of primaquine in patients withP. Vivax malaria. Eur J Clin Pharmacol 31, 205–210 (1986). https://doi.org/10.1007/BF00606660
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00606660