Abstract
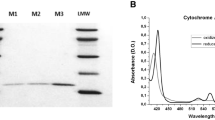
Human cyclo-oxygenase-1 (hCOX-1) and-2 were expressed in stable transfected COS A.2 cells and in insect cells using a Sf9 baculovirus expression system. Inhibition of COX activity was examined using both whole cell and microsomal assays. Ibuprofen, naproxen, 6-MNA, diclofenac and indomethacin were selective for hCOX-1 or were equipotent inhibitors for COX-1 and COX-2. Piroxicam was equally inhibitory for both enzymes in the whole cell assay while it preferentially inhibited hCOX-2 in the microsomal assay. However, maximal inhibition of hCOX-2 by piroxica plateaued at 60%. Nimesulide was equipotent in the whole-cell assay but was five-fold selective for hCOX-2 in the microsomal assay. Meloxicam preferentially inhibited hCOX-2 in the whole cell assay at concentrations of 0.01 to 1 μmol/L but was an equipotent inhibitor of both enzymes at higher concentrations. In the microsomal assay, meloxicam exhibited high selectivity for hCOX-2 (75-fold). The preferential inhibition of hCOX-2 by meloxicam may explain the favourable gastrointestinal profile observed for meloxicam compared with other NSAIDs.
Similar content being viewed by others
References
Vane JR. Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nature. 1971;231:232–7.
Fu JY, Masferrer JL, Seibert K, Raz A, Needleman P. The induction and suppression of prostaglandin H2 synthase (cyclooxygenase) in human monocytes. J Biol Chem. 1990;265:16737–40.
Xie W, Chipman JG, Robertson DL, Erikson RL, Simmons DL. Expression of a mitogen-responsive gene encoding prostaglandin synthase is regulated by mRNA splicing. Proc Natl Acad Sci USA. 1991;88:2692–6.
O’Banion MK, Sadowski HB, Winn V, Young DA. A serum- and glucocorticoid-regulated 4-kilobase mRNA encodes a cyclooxygenase-related protein. J Biol Chem. 1991;266:23261–7.
Simmons DL, Xie W, Chipman JG, Evett GE. Multiple cyclooxygenases: Cloning of a mitogen-inducible form. In: Bailey JM, ed. Prostaglandins, Leukotrienes, Lipoxins and PAF. New York: Plenum Press; 1991:67–78.
Jones DA, Carlton DP, McIntyre TM, Zimmerman GA, Prescott SM. Molecular cloning of human prostaglandin endoperoxide synthase type II and demonstration of expression in, response to cytokines. J Biol Chem. 1993;268:9049–54.
Kujubu DA, Fletcher BS, Varnum BC, Lim RW, Herschman HR. TIS10, a phorbol ester tumor promoter-inducible mRNA from Swiss 3T3 cells, encodes a novel prostaglandin synthase/cyclo-oxygenase homologue. J Biol Chem., 1991;266:12866–72.
Lee SH, Soyoola E, Chanmugam P et al. Selective expression of mitogen-inducible cyclooxygenase in macrophages stimulated with lipopolysaccharide. J Biol Chem. 1992;267:25934–8.
Masferrer JL, Zweifel BS, Manning PT et al. Selective inhibition of inducible cyclooxygenase 2 in vivo is antiinflammatory and nonulcerogenic. Proc Natl Acad Sci USA. 1994;91:3228–32.
Sano H, Hla HM, Maier JA et al. In vivo cyclooxygenase expression in synovial tissues of patients with rheumatoid arthritis and osteoarthritis and rats with adjuvant and streptococcal cell wall arthritis. J Clin Invest. 1992;89:97–108.
Engelhardt G, Homma D, Schlegel K, Utzmann R, Schnitzler C. Anti-inflammatory, analgesic, antipyretic and related properties of meloxicam, a new nonsteroidal anti-inflammatory agent with favourable gastrointestinal tolerance. Inflamm Res. 1995;44:423–33.
Engelhardt G, Homma D, Schnitzler C. Meloxicam: A potent inhibitor of adjuvant arthritis in the lewis rat. Inflamm Res.. 1995;44:548–55.
Ormancy A, Horeau A. Molecular structure and estrogen activity XVI. Influence of the distance of functional groups with the allenolique series. Bull Soc Chim Fr. 1955;XI:962–8.
Holford NHG, Sheiner LB. Pharmacokinetic and pharmacodynamic modelling in vivo. Crit Rev Bioeng. 1981;5:273–322.
Holford NHG, Sheiner LB. Kinetics of pharmacologic response. Pharmacol Ther.. 1982;16:143–66.
Mitchell JA, Akarasereenont P, Thiermann C, Flower RJ, Vane JR. Selectivity of nonsteroidal antiinflammatory drugs as inhibitors of constitutive and inducible cyclooxygenase. Proc Natl Acad Sci USA. 1993;90:11693–7.
Grossman CJ, Wiseman J, Lucas FS, Trevethick MA, Birch PJ. Inhibition of constitutive and inducible cyclooxygenase activity in human platelets and mononuclear cells by NSAIDs and COX-2 inhibitors.Inflamm Res.. 1995;44:253–7.
Engelhardt G, Pariet M. Meloxicam: A new NSAID with an improved safety profile through preferential inhibition of COX-2. Rheumatol Eur. 1995;24(suppl 3):D203.
Barnett J, Chow J, Ives D et al. Purification, characterization and selective inhibition of human prostaglandin G/H synthase 1 and 2 expressed in the baculovirus system. Biochim Biophys Acta. 1994;1209:130–9.
O’Neill GP, Mancini JA, Kargman S et al. Overexpression of human prostaglandin G/H synthase-1 and-2 by recombinant vaccina virus: Inhibition by nonsteroidal antiinflammatory drugs and biosynthesis of 15-hydroxyeicosatetrenoic acid. Mol Pharmacol. 1995;45:245–54.
Glaser K, Sung ML, O’Neil K et al. Etodolac selectively inhibits human prostaglandin G/H synthase 2 (PGHS-2) versus human PGHS-1. Eur J Pharmacol. 1995;281:107–11.
Turck D, Busch U, Heinzel G, Narjes H. Clinical pharmacokinetics of meloxicam. Eur J Rheumatol Inflamm. 1996;15:23–30.
Distel M, Mueller C, Blumkhi E. Global analysis of safety of a new NSAID, meloxicam. Rheumatol Eur. 1995;24(suppl 3):E259.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Churchill, L., Graham, A.G., Shih, CK. et al. Selective inhibition of human cyclo-oxygenase-2 by meloxicam. Inflammopharmacology 4, 125–135 (1996). https://doi.org/10.1007/BF02735467
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02735467