Abstract
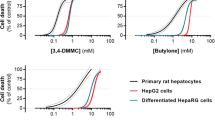
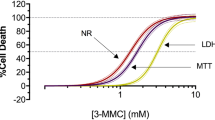
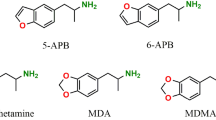
The amphetamine designer drugs 3,4-methylenedioxymethamphetamine (MDMA or “ecstasy”) and its N-demethylated analogue 3,4-methylenedioxyamphetamine (MDA or “love”) have been extensively used as recreational drugs of abuse. MDA itself is a main MDMA metabolite. MDMA abuse in humans has been associated with numerous reports of hepatocellular damage. Although MDMA undergoes extensive hepatic metabolism, the role of metabolites in MDMA-induced hepatotoxicity remains unclear. Thus, the aim of the present study was to evaluate the effects of MDA and α-methyldopamine (α-MeDA), a major metabolite of MDA, in freshly isolated rat hepatocyte suspensions. The cells were incubated with MDA or α-MeDA at final concentrations of 0.1, 0.2, 0.4, 0.8, or 1.6 mM for 3 h. The toxic effects induced following incubation of hepatocyte suspensions with these metabolites were evaluated by measuring cell viability, the extent of lipid peroxidation, levels of glutathione (GSH) and glutathione disulfide (GSSG), the formation of GSH conjugates, and the activities of GSSG reductase (GR), GSH peroxidase (GPX), and GSH S-transferase (GST). MDA induced a concentration- and time-dependent GSH depletion, but had a negligible effect on lipid peroxidation, cell viability, or on the activities of GR, GPX, and GST. In contrast, α-MeDA (1.6 mM, 3 h) induced a marked depletion of GSH accompanied by a loss on cell viability, and decreases in GR, GPX and GST activities, although no significant effect on lipid peroxidation was found. For both metabolites, GSH depletion was not accompanied by increases in GSSG levels; rather, 2-(glutathion-S-yl)-α-MeDA and 5-(glutathion-S-yl)-α-MeDA were identified by HPLC-DAD/EC within cells incubated with MDA or α-MeDA. The results provide evidence that one of the early consequences of MDMA metabolism is a disruption of thiol homeostasis, which may result in loss of protein function and the initiation of a cascade of events leading to cellular damage.







Similar content being viewed by others
References
Anderson ME (1985) Determination of glutathione and glutathione disulfide in biological samples. Methods Enzymol 113:548–555
Armstrong RN (1991) Glutathione S-transferases: reaction mechanism, structure, and function. Chem Res Toxicol 4:131–140
Bai F, Lau SS, Monks TJ (1999) Glutathione and N-acetylcysteine conjugates of α-methyldopamine produce serotoninergic neurotoxicity: possible role in methylenedioxyamphetamine-mediated neurotoxicity. Chem Res Toxicol 12:1150–1157
Bai F, Jones DC, Lau SS, Monks TJ (2001) Serotonergic neurotoxicity of 3,4-(±)-methylenedioxyamphetamine and 3,4-(±)-methylenedioxymethamphetamine (Ecstasy) is potentiated by inhibition of γ-glutamyl transpeptidase. Chem Res Toxicol 14:863–870
Battaglia G, Yeh SY, O’Hearn E, Molliver ME, Kuhar MJ, Sousa EBD (1987) 3,4-Methylenedioxymethamphetamine and 3,4-methylenedioxyamphetamine destroy serotonin terminals in rat brain: quantification of neurodegeneration by measurement of [3H]paroxetine-labeled serotonin uptake sites. J Pharmacol Exp Ther 242:911–916
Beitia G, Cobreros A, Sainz L, Cenarruzabeitia E (1999) 3,4-Methylenedioxymethamphetamine (ecstasy)-induced hepatotoxicity: effect on cytosolic calcium signals in isolated hepatocytes. Liver 19:234–241
Ben-Shachar D, Riederer P, Youdim MB (1991) Iron-melanin interaction and lipid peroxidation: implications for Parkinson’s disease. J Neurochem 57:1609–1614
Bindoli A, Rigobello MP, Galzigna L (1989) Toxicity of aminochromes. Toxicol Lett 48:3–20
Bindoli A, Rigobello MP, Deeble DJ (1992) Biochemical and toxicological properties of the oxidation products of catecholamines. Free Radic Biol Med 13:391–405
Bolton JL, Trush MA, Penning TM, Dryhurst G, Monks TJ (2000) Role of quinones in toxicology. Chem Res Toxicol 13:135–160
Carstam R, Brinck C, Hindemith-Augustsson A, Rorsman H, Rosengren E (1991) The neuromelanin of the human substancia nigra. Biochim Biophys Acta 1097:152–160
Carvalho F, Remião F, Soares ME, Catarino R, Queiroz G, Bastos ML (1997) d-Amphetamine-induced hepatotoxicity: possible contribution of catecholamines and hyperthermia to the effect studied in isolated rat hepatocytes. Arch Toxicol 71:429–436
Carvalho M, Carvalho F, Bastos ML (2001) Is hyperthermia the triggering factor for hepatotoxicity induced by 3,4-methylenedioxymethamphetamine (ecstasy)? An in vitro study using freshly isolated mouse hepatocytes. Arch Toxicol 74:789–793
Carvalho M, Carvalho F, Remião F, Pereira MdL, Pires-das-Neves R, Bastos MdL (2002a) Effect of 3,4-methylenedioxymethamphetamine (“ecstasy”) on body temperature and liver antioxidant status in mice: influence of ambient temperature. Arch Toxicol 76:166–172
Carvalho M, Hawksworth G, Milhazes N, Borges F, Monks T, Fernandes E, Carvalho F, Bastos ML (2002b) Role of metabolites in MDMA (ecstasy)-induced nephrotoxicity: an in vitro study using rat and human renal proximal tubular cells. Arch Toxicol 76:581–588
Cho AK, Hiramatsu M, Distefano EW, Chang AS, Jenden DJ (1990) Stereochemical differences in the metabolism of 3,4-methylenedioxymethamphetamine in vivo and in vitro: a pharmacokinetic analysis. Drug Metab Dispos 18:686–691
DeLeve LD, Kaplowitz N (1991) Glutathione metabolism and its role in hepatotoxicity. Pharmacol Ther 52:287–305
Di Mascio P, Murphy ME, Sies H (1991) Antioxidant defense systems: the role of carotenoids, tocopherols, and thiols. Am J Clin Nutr 53:194S–200S
Dykhuizen RS, Brunt PW, Atkinson P, Simpson JG, Smith CC (1995) Ecstasy induced hepatitis mimicking viral hepatitis. Gut 36:939–941
Ellis A, Wendon J, Portmann B, Williams R (1996) Acute liver damage and ecstasy ingestion. Gut 38:454–458
Fineschi V, Centini F, Mazzeo E, Turillazzi E (1999) Adam (MDMA) and Eve (MDA) misuse: an immunohistochemical study on three fatal cases. Forensic Sci Int 104:65–74
Hegadoren KM, Baker GB, Bourin M (1999) 3,4-Methylenedioxy analogues of amphetamine: defining the risks to humans. Neurosci Biobehav Rev 23:539–553
Hegedus ZL (2000) The probable involvement of soluble and deposited melanins, their intermediates and the reactive oxygen side-products in human diseases and aging. Toxicology 145:85–101
Henry JA, Jeffreys KJ, Dawling S (1992) Toxicity and deaths from 3,4-methylenedioxymethamphetamine (“ecstasy”). Lancet 340:384–387
Hiramatsu M, Kumagai Y, Unger SE, Cho AK (1990) Metabolism of methylenedioxymethamphetamine: formation of dihydroxymethamphetamine and a quinone identified as its glutathione adduct. J Pharmacol Exp Ther 254:521–527
Khakoo S, Coles C, Armstrong J, Barry R (1995) Hepatotoxicity and accelerated fibrosis following 3,4-methylenedioxymethamphetamine (“ecstasy”) usage. J Clin Gastroenterol 20:244–247
Kreth K-P, Kovar K-A, Schwab M, Zanger UM (2000) Identification of the human cytochromes P450 involved in the oxidative metabolism of “ecstasy”-related designer drugs. Biochem Pharmacol 59:1563–1571
Kumagai Y, Lin LY, Schmitz DA, Cho AK (1991) Hydroxyl radical mediated demethylenation of (methylenedioxy)phenyl compounds. Chem Res Toxicol 4:330–334
Lim HK, Foltz RL (1988) In vivo and in vitro metabolism of 3,4-(methylenedioxy)methamphetamine in the rat: identification of metabolites using an ion trap detector. Chem Res Toxicol 1:370–378
Mannervik B, Danielson UH (1988) Glutathione transferases-structure and catalytic activity. CRC Crit Rev Biochem 23:283–337
Marquardt GM, DiStefano V, Ling LL (1978) Metabolism of β-3,4-methylenedioxyamphetamine in the rat. Biochem Pharmacol 27:1503–1505
Miller RT, Lau SS, Monks TJ (1995) Metabolism of 5-(glutathion-S-yl)-α-methyldopamine following intracerebroventricular administration to male Sprague-Dawley rats. Chem Res Toxicol 8:634–641
Miller RT, Lau SS, Monks TJ (1996) Effects of intracerebroventricular administration of 5-(glutathion-S-yl)-α-methyldopamine on brain dopamine, serotonin, and norepinephrine concentrations in male Sprague-Dawley rats. Chem Res Toxicol 9:457–465
Miller RT, Lau SS, Monks TJ (1997) 2,5-bis-(glutathion-S-yl)-α-methyldopamine, a putative metabolite of (±)-3,4-methylenedioxyamphetamine, decreases brain serotonin concentrations. Eur J Pharmacol 323:173–180
Milroy CM, Clark JC, Forrest ARW (1996) Pathology of deaths associated with “ecstasy” and “eve” misuse. J Clin Pathol 49:149–153
Moldéus P, Hogberg J, Orrenius S (1978) Isolation and use of liver cells. Methods Enzymol 52:60–71
Monks TJ, Lau SS (1992) Toxicology of quinone-thioethers. Crit Rev Toxicol 22:243–270
Ommen BV, Ploemen JHTM, Bogaards JJP, Monks TJ, Lau SS, Bladeren PJV (1991) Irreversible inhibition of rat glutathione S-transferase 1-1 by quinones and their glutathione conjugates. Structure–activity relationship and mechanism. Biochem J 276:661–666
Ploemen JH, Ommen BV, Bladeren PJV (1991) Irreversible inhibition of human glutathione S-transferase isoenzymes by tetrachloro-1,4-benzoquinone and its glutathione conjugate. Biochem Pharmacol 41:1665–1669
Ploemen JH, Ommen BV, Haan AD, Venekamp JV, Bladeren PJV (1994) Inhibition of human glutathione S-transferases by dopamine, α-methyldopa and their 5-S-glutathionyl conjugates. Chem Biol Interact 90:87–99
Reed DJ (1990) Glutathione: toxicological implications. Annu Rev Toxicol 30:603–631
Remião F, Carmo H, Carvalho FD, Bastos ML (1999) Inhibition of glutathione reductase by isoproterenol oxidation products. J Enzyme Inhib 15:47–61
Remião F, Carvalho M, Carmo H, Carvalho FD, Bastos ML (2002) Cu2+-induced isoproterenol oxidation into isoprenochrome in adult rat calcium-tolerant cardiomyocytes. Chem Res Toxicol 15:861–869
Ricaurte GA, Yuan J, McCann UD (2000) (±)3,4-Methylenedioxymethamphetamine (“ecstasy”)-induced serotonin neurotoxicity: studies in animals. Neuropsychobiology 42:5–10
Simpson DL, Rumack BH (1981) Methylenedioxyamphetamine. Clinical description of overdose, death, and review of pharmacology. Arch Intern Med 141:1507–1509
Spencer JP, Jenner P, Daniel SE, Lees AJ, Marsden DC, Halliwell B (1998) Conjugates of catecholamines with cysteine and GSH in Parkinson’s disease: possible mechanisms of formation involving reactive oxygen species. J Neurochem 71:2112–2122
van Bladeren PJ (2000) Glutathione conjugation as a bioactivation reaction. Chem Biol Interact 129:61–76
Varela-Rey M, Montiel-Duarte C, Beitia G, Cenarruzabeitia E, Iraburu MJ (1999) 3,4-Methylenedioxymethamphetamine (“ecstasy”) stimulates the expression of α1 (I) procollagen mRNA in hepatic stellate cells. Biochem Biophys Res Commun 259:678–682
Walubo A, Seger D (1999) Fatal multi-organ failure after suicidal overdose with MDMA, “ecstasy”: case report and review of the literature. Hum Exp Toxicol 18:119–125
Zhang F, Dryhurst G (1994) Effects of l-cysteine on the oxidation chemistry of dopamine: new reaction pathways of potential relevance to idiopathic Parkinson’s disease. J Med Chem 37:1084–1098
Acknowledgements
This work was supported by PhD grants from Fundação para a Ciência e Tecnologia (Praxis XXI/BD/20087/99 and Praxis XXI/BD/18520/98) and Programa Operacional Ciência, Tecnologia e Inovação (POCTI), Portugal, in co-participation with Fonds Européen de Développement Régional (FEDER) (project POCTI/36099/FCB/2000). The experiments are declared to comply with the current laws of Portugal.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carvalho, M., Milhazes, N., Remião, F. et al. Hepatotoxicity of 3,4-methylenedioxyamphetamine and α-methyldopamine in isolated rat hepatocytes: formation of glutathione conjugates. Arch Toxicol 78, 16–24 (2004). https://doi.org/10.1007/s00204-003-0510-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-003-0510-7