Abstract
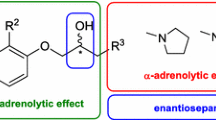
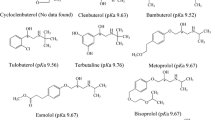
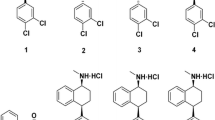
We separated and characterized the enantiomers of bambuterol (5-[-(tert-butylamino)-1-hydroxyethyl]-m-phenylene-bis(dimethylcarbamate) hydrochloride), which is used in racemic form as a prodrug of terbutaline, a β2-adrenoceptor agonist. The enantioseparation was attempted on several chiral HPLC columns, and the most effective separation was achieved on the amylose-based Chiralpak AD column. Since in vivo conversion of bambuterol into terbutaline involves hydrolysis by butyrylcholinesterase (EC 3.1.1.8), we studied the reaction of enantiomers with eight human BChE variants. Both enantiomers inhibited all studied BChE variants; however, the rate of inhibition with the (R)-enantiomer was about five times faster than with the (S)-enantiomer. (R)-bambuterol inhibition rate constants for homozygous usual (UU), fluoride-resistant (FF) or atypical (AA) variant ranged from 6.4 to 0.11 min-1μM-1. The inhibition rates for heterozygotes were between the respective constants for the corresponding homozygotes.







Similar content being viewed by others
References
Bush RK, Georgitis JW (1997) (eds) Handbook of asthma and rhinitis. Blackwell, Abingdon
Waldeck B (2002) Eur J Pharmacol 445:1–12
Tunek A, Svensson LÅ (1988) Drug Metab Dispos 16:759–764
Svensson LÅ, Tunek A (1988) Drug Metab Rev 19:165–194
Tunek A, Hjertberg E, Viby-Mogensen J (1991) Biochem Pharmacol 41: 345–348
Kovarik Z, Simeon-Rudolf V (2004) J Enzyme Inhib Med Chem 19:113–117
Kovarik Z (1999) Period Biol 101:7–15
Primo-Parmo SL, Bartels CF, Wiersema B, Van Der Spek AFL, Innis JW, La Du BN (1996) Am J Hum Genet 58:52–64
Evans RT (1986) Crit Rev Clin Lab Sci 23:35–64
Bartels CF, Jensen FS, Lockridge O, Van Der Spek AFL, Rubinstein HM, Lubrano T, La Du BN (1992) Am J Hum Genet 50:1086–1103
Pálmarsdóttir S, Mathiasson L, Jönson JA, Edholm LE (1997) J Chromatogr B 688:127–134
Bartolinčić A, Drušković V, Šporec A, Vinković V (2005) J Pharm Biomed Anal 36:1003–1010
Matlin SA, Tirtan ME, Cass QB, Boyd DR (1996) Chirality 8:147–152
Kim IW, Ryu JK, Ahn SD, Park JH, Lee KP, Ryoo JJ, Hyun MH, Okamoto Y, Yamamoto C, Carr PW (2003) Bull Korean Chem Soc 24:239–242
Tan W, Cheng JL (2005) US Patent No. 2005/0171197
Kovarik Z, Simeon-Rudolf V (2003) Arh Hig Rada Toksikol 54:239–244
Simeon-Rudolf V, Evans RT (2001) Acta Pharm 51:289–296
Ellman GL, Courtney KD, Andres V Jr, Featherstone RM (1961) Biochem Pharmacol 7:88–95
Sajonz P, Natishan TK, Antia FD, Frenette R (2005) J Chromatogr A 1089:135–141
Gazi I, Kontrec D, Lesac A, Vinković V (2005) Tetrahedron: Asymm 16:1175–1182
Zhang T, Kientzy C, Franco P, Ohnishi A, Kagamira Y, Kurosawa H (2005) J Chromatogr A 1075:65–75
Ghanem A, Aboul-Enein HY (2005) Anal Chim Acta 548:26–32
Brown HC, Ramachandran V (1992) Acc Chem Res 25:16–24
Lockridge O (1990) Pharmac Ther 47:35–60
Nicolet Y, Lockridge O, Masson P, Fontecilla-Camps JC, Nachon F (2003) J Biol Chem 278:41141–41147
Masson P, Adkins S, Gouet P, Lockridge O (1993) J Biol Chem 268:14329–14341
Simeon-Rudolf V, Kovarik Z, Škrinjarić-Špoljar M, Evans RT (1999) Chem-Biol Inter 199–120:159–164
Prester L, Simeon V (1991) Biochem Pharmacol 42: 2313–2316
Sitar DS (1996) Clin Pharmacokinet 31:246–256
Nyberg L, Rosenborg J, Weibull E, Jönsson S, Kennedy BM, Nilsson M (1998) Br J Clin Pharmacol 45:471–478
Ahlström H, Alvero J, Alvero R, Espos R, Fajutrao L, Herrera J, Kjellman B, Kubista J, Leviste C, Meyer P, Oldæus G, Siricururat A, Vichyanond P, Wettrell G, Wong E, Laxmyr L, Nyberg L, Olsson H, Weibull E, Rosenborg J (1999) Br J Clin Pharmacol 48:299–308
Rosenborg J, Larsson P, Nyberg L (2000) Br J Clin Pharmacol 49:199–206
Kovarik Z, Radi Z, Grgas B, Škrinjari-Špoljar M, Reiner E, Simeon-Rudolf V (1999) Biochim Biophys Acta 1433:261–271
Kovarik Z, Bosak A, Šinko G, Latas T (2003) Croat Chem Acta 76:63–67
Kovarik Z, Radić Z, Berman HA, Simeon-Rudolf V, Reiner E, Taylor P (2003) Biochem J 373:33–40
Bang U, Nyberg L, Rosenborg J, Viby-Mogensen J (1998) Br J Clin Pharmacol 45:479–484
Acknowledgements
We wish to thank AstraZeneca for providing us with racemic bambuterol as well as Dr. R.T. Evans and Dr. O. Lockridge for providing us with rare serum specimens. This work was supported by the Ministry of Science, Education and Sports of the Republic of Croatia.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found at http://dx.doi.org/10.1007/s00216-006-0864-9
Rights and permissions
About this article
Cite this article
Gazić, I., Bosak, A., Šinko, G. et al. Preparative HPLC separation of bambuterol enantiomers and stereoselective inhibition of human cholinesterases. Anal Bioanal Chem 385, 1513–1519 (2006). https://doi.org/10.1007/s00216-006-0566-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-006-0566-3