Abstract
Rationale
Preliminary in vitro findings indicated that the novel anxiolytic drug, deramciclane is a substrate for the cytochrome P 450 (CYP) 3A4 isoenzyme. Moreover, its co-administration with buspirone, another anxiolytic drug, is likely in clinical practice.
Objectives
The primary objective of the present study was to evaluate the in vivo effects of deramciclane on CYP3A4 activity as measured by buspirone pharmacokinetics. The secondary objective was to study the possible pharmacodynamic interaction between these two anxiolytic drugs.
Methods
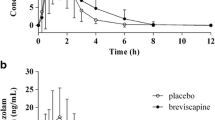
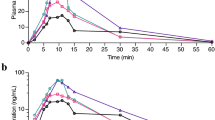
Sixteen healthy subjects received 60 mg deramciclane or matched placebo for 8 days in this randomized, double-blind, cross-over study. On day 8 of both phases, the subjects received a 20-mg single dose of buspirone. Buspirone and its active metabolite, 1-pyrimidylpiperazine (1-PP), concentrations were measured for 24 h. Pharmacodynamic testing and measurement of plasma prolactin concentrations were carried out on day 7 and day 8 to assess the pharmacodynamic consequences of deramciclane and buspirone co-administration.
Results
Repeated administration of deramciclane had no effect on CYP3A4 activity as measured by buspirone pharmacokinetics. However, deramciclane administration caused an inhibition of the further, not CYP3A4-dependent, metabolism of 1-PP as evidenced by 84% increase in the AUC (P<0.001) and 20% increase in the elimination half-life (P=0.0012) of 1-PP. Deramciclane did not potentiate the buspirone-induced increase in prolactin secretion. No significant differences were found in the psychomotoric testing or the subjective maximum sedation between the deramciclane phase and the placebo phase, either before or after buspirone administration. Of 16 subjects, 5 experienced dizziness during both study phases.
Conclusion
Deramciclane does not inhibit CYP3A4 activity as measured by buspirone pharmacokinetics, and there were no indications of relevant pharmacodynamic interaction after multiple doses of deramciclane and a single dose of buspirone.


Similar content being viewed by others
References
Bond A, Lader M (1974) The use of analogue scales in rating subjective feelings. Br J Med Psychol 47:211–218
Bond A, Lader M, Shrotriya R (1983) Comparative effects of a repeated dose regime of diazepam and buspirone on subjective ratings, psychological tests and the EEG. Eur J Clin Pharmacol 24:463–467
Bridge MW, Marvin G, Thompson CE, Sharma A, Jones DA, Kendall MJ (2001) Quantifying the 5 HT1A agonist action of buspirone in man. Psychopharmacology 158:224–249
Cohn JB, Wilcox CS (1986) Low-sedation potential of buspirone compared with alprazolam and lorazepam in the treatment of anxious patients: a double-blind study. J Clin Psychiatry 47:409–412
Dinan TG, Scott LV, Thakore J, Naesdal J, Keeling PW (2001) Impact of cortisol on buspirone stimulated prolactin release: a double-blind placebo-controlled study. Psychoneuroendocrinology 26:751–756
FDA CDER (1999) Guidance for industry: in vivo drug metabolism/drug interaction studies: study design, data analysis, and recommendations for dosing and labeling [online]. Available: http://www.fda.gov/cder
Gacsalyi I, Gigler G, Szabados T, Kovacs, Vasar E, Männistö PT (1996) Different antagonistic activity of deramciclane (EGIS-3886) on peripheral and central 5-HT2 receptors. Pharm Pharmacol Lett 6:82–85
Gammans RE, Mayol RF, LaBudde JA (1986) Metabolism and disposition of buspirone. Am J Med 80:41–51
Ghoneim MM, Hinrichs JV, Mewaldt SP (1984) Dose–response analysis of the behavioral effects of diazepam: I. Learning and Memory. Psychopharmacology 82:291–295
Jajoo HK, Mayol RF, LaBudde JA, Blair IA (1989) Metabolism of the antianxiety drug buspirone in human subjects. Drug Metab Dispos 17:634–640
Kanerva H, Kilkku O, Heinonen E, Helminen A, Rouru J, Tarpila S, Scheinin M, Huupponen R, Klebovich I, Drabant S, Urtti A (1999a) The single dose pharmacokinetics and safety of deramciclane in healthy male volunteers. Biopharm Drug Dispos 20:327–334
Kanerva H, Kilkku O, Helminen A, Rouru J, Scheinin M, Huupponen R, Klebovich I, Drabant S, Urtti A (1999b) Pharmacokinetics and safety of deramciclane during multiple oral dosing. Int J Clin Pharmacol Ther 37:589–597
Kanerva H, Vilkman H, Nagren K, Kilkku O, Kuoppamaki M, Syvalahti E, Hietala J (1999c) Brain 5-HT2A receptor occupancy of deramciclane in humans after a single oral administration—a positron emission tomography study. Psychopharmacology 145:76–81
Kerns EH, Rourick RA, Volk KJ, Lee MS (1997) Buspirone metabolite structure profile using a standard liquid chromatographic-mass spectrometric protocol. J Chromatogr B Biomed Sci Appl. 698:133–145
Kivistö KT, Lamberg TS, Kantola T, Neuvonen PJ (1997) Plasma buspirone concentrations are greatly increased by erythromycin and itraconazole. Clin Pharmacol Ther 62:348–354
Lamberg TS, Kivisto KT, Laitila J, Martensson K, Neuvonen PJ (1998a) The effect of fluvoxamine on the pharmacokinetics and pharmacodynamics of buspirone. Eur J Clin Pharmacol 54:761–766
Lamberg TS, Kivisto KT, Neuvonen PJ (1998b) Effects of verapamil and diltiazem on the pharmacokinetics and pharmacodynamics of buspirone. Clin Pharmacol Ther 63:640–645
Meltzer HY, Maes M (1994) Effects of buspirone on plasma prolactin and cortisol levels in major depressed and normal subjects. Biol Psychiatry 35:316–323
Naukkarinen H (2001) Deramciclane in the treatment of patients with generalized anxiety disorder: a randomized, double-blind, placebo controlled, dose-finding study. Eur Neuropsychopharmacol 11[Suppl]:S301
Pälvimäki EP, Majasuo H, Kuoppamäki M, Männistö PT, Syvälahti E, Hietala J (1998) Deramciclane, a putative anxiolytic drug, is a serotonin 5-HT2C receptor inverse agonist but fails to induce 5-HT2C receptor down-regulation. Psychopharmacology 136:99–104
Seidel WF, Cohen SA, Wilson L, Dement WC (1985) Effects of alprazolam and diazepam on the daytime sleepiness of non-anxious volunteers. Psychopharmacology 87:194–197
Stone B (1984) Pencil and paper tests—sensitivity to psychotropic drugs. Br J Clin Pharmacol 18:15–20
Yocca FD (1990) Neurochemistry and neurophysiology of buspirone and gepirone: interactions at presynaptic and postsynaptic 5-HT1A receptors. J Clin Psychopharmacol 10[Suppl]:6S–12S
Acknowledgements
This study was sponsored financially by Orion Pharma.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Laine, K., Ahokoski, O., Huupponen, R. et al. Effect of the novel anxiolytic drug deramciclane on the pharmacokinetics and pharmacodynamics of the CYP3A4 probe drug buspirone. Eur J Clin Pharmacol 59, 761–766 (2003). https://doi.org/10.1007/s00228-003-0674-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-003-0674-3