Abstract
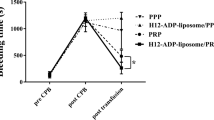
Severe thrombocytopenia frequently occurs in patients receiving chemotherapy and in patients with autoimmune disorders. Thrombocytopenia is associated with bleeding, which may be serious and life threatening1,2,3. Current treatment strategies for thrombocytopenia may require transfusion of allogeneic platelets, which is associated with serious drawbacks4. These include the occurrence of anti-platelet antibodies, which may result in refractoriness to further platelet transfusions, and the potential risk of transfer of blood-borne diseases5,6. Therefore, we have recently developed a platelet substitute product (Synthocytes), which is composed of human albumin microcapsules with fibrinogen immobilized on their surface. Here we show that the intravenous administration of these microcapsules not only corrects the prolonged bleeding time in rabbits rendered thrombocytopenic either by anti-platelet antibodies or by chemotherapy, but also reduces bleeding from surgical wounds inflicted in the abdominal skin and musculature. No potential systemic prothrombotic effect of the microcapsules was observed in a model of rabbit venous thrombosis. As for the mechanism of action, experiments with normal and thrombocytopenic human blood in an endothelial cell matrix-coated perfusion chamber demonstrated an interaction between the fibrinogen-coated albumin microcapsules and native platelets. It was shown that the fibrinogen-coated albumin microcapsules could facilitate platelet adhesion to endothelial cell matrix and correct the impaired formation of platelet aggregates in relatively platelet-poor blood. This study indicates that fibrinogen-coated albumin microcapsules can act to improve primary hemostasis under thrombocytopenic conditions and may eventually be a promising agent for prophylaxis and treatment of bleeding in patients with severe thrombocytopenia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Roy, A.J., Jaffe, N. & Djerassi, I. Prophylactic platelet transfusions in children with acute leukemia. A dose response study. Transfusion 13, 283–290 (1973).
Rebulla, P. et al. The threshold for prophylactic platelet transfusions in adults with acute myeloid leukemia. N. Engl. J. Med. 337, 1870–1875 (1997).
National Institute of Health Consensus Conference. Platelet transfusion therapy.Transfus. Med. Rev. 1, 195 –200 (1987).
Slichter, S.J. in Hematology: Basic Principles and Practice 2nd edn. (eds. Hoffman, R. et al. ) 419–421 (Churchill Livingstone, New York, 1995).
The Trial to Reduce Alloimmunization to Platelets Study Group. Leucocyte reduction and ultraviolet B irradiation of platelets to prevent alloimmunization and refractoriness to platelet transfusions. N. Engl. J. Med. 337, 1861–1869 (1997).
Dodd, R.Y. The risk of transfusion-transmitted infection. N. Engl. J. Med. 327, 419–421 ( 1992).
Yen, R.C.K., & Ho, T.W.C. Blajchman M.A. A novel approach to correcting the bleeding time in thrombocytopenic rabbits. Transfusion 35, 41S (1995).
Rodgers, R.P. & Levin, J. A critical reappraisal of the bleeding time. Semin. Thromb. Hemost. 16, 1– 20 (1990).
Evatt, B.L., Levin, J. & Algazy, K.M. Partial purification of thrombopoietin from the plasma of thrombocytopenic rabbits. Blood 54, 377 –388 (1979).
Blajchman, M.A. & Lee, D.H. The thrombocytopenic rabbit bleeding time model to evaluate the in vivo hemostatic efficacy of platelets and platelet substitutes. Transf. Med. Rev. 11, 99–105 (1997).
Kuter, D.J. & Rosenberg, R.D. The reciprocal relationship of thrombopoietin (c-Mpl ligand) to changes in the platelet mass during busulfan-induced thrombocytopenia in the rabbit. Blood 85, 2710–2730 (1995).
Blajchman, M.A. et al. Shortening of the bleeding time in rabbits by hydrocortisone caused inhibition of prostacyclin generation by the vessel wall. J. Clin. Invest. 63, 1026–1035 (1979).
Benedict, C.R. et al. New variant of human tissue plasminogen activator (TPA) with enhanced efficacy and lower incidence of bleeding compared with recombinant human TPA. Circulation 92, 3032– 3040 (1995).
Levi, M., Biemond, B.J., van Zonneveld, A.J., ten Cate, J.W. & Pannekoek, H. : Inhibition of plasminogen activator inhibitor 1 (PAI-1) activity results in promotion of endogenous thrombolysis and inhibition of thrombus extension in models of experimental thrombosis. Circulation 85, 305–312 (1992).
Sakariassen, K.S., Aarts, P.A., de Groot, P.G., Houdijk, W.P. & Sixma, J.J. A perfusion chamber developed to investigate platelet interaction in flowing blood with human vessel wall cells, their extracellular matrix, and purified components. J. Lab. Clin. Med. 102, 522–535 ( 1983).
Zwaginga, J.J. et al. Thrombogenicity of vascular cells. Comparison between endothelial cells isolated from different sources and smooth muscle cells and fibroblasts. Arteriosclerosis 10, 437– 448 (1990).
van Breugel, H.F., de Groot, P.G., Heethaar, R.M. & Sixma, J.J. Role of plasma viscosity in platelet adhesion. Blood 80, 953–959 (1992).
Saelman, E.U.M. et al. Aggregate formation is more strongly inhibited at high shear rates by dRGDW, a synthetic RGD-containing peptide. Arterioscler. Thromb. 13, 1164–1170 ( 1993).
Acknowledgements
The assistance of A. van de Wardt is acknowledged. M.L. is a fellow of the Royal Dutch Academy of Arts and Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Levi, M., Friederich, P., Middleton, S. et al. Fibrinogen-coated albumin microcapsules reduce bleeding in severely thrombocytopenic rabbits. Nat Med 5, 107–111 (1999). https://doi.org/10.1038/4795
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/4795
This article is cited by
-
Platelet Contributions to Trauma-Induced Coagulopathy: Updates in Post-injury Platelet Biology, Platelet Transfusions, and Emerging Platelet-Based Hemostatic Agents
Current Trauma Reports (2019)
-
Engineering blood cells and proteins as blood substitutes: A short review
Biotechnology and Bioprocess Engineering (2007)
-
New strategy of platelet substitutes for enhancing platelet aggregation at high shear rates: cooperative effects of a mixed system of fibrinogen γ-chain dodecapeptide- or glycoprotein Ibα-conjugated latex beads under flow conditions
Journal of Artificial Organs (2006)