Abstract
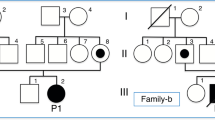
Primary systemic carnitine deficiency (SCD; OMIM 212140) is an autosomal recessive disorder characterized by progressive cardiomyopathy, skeletal myopathy, hypoglycaemia and hyperammonaemia1,2,3. SCD has also been linked to sudden infant death syndrome4. Membrane-physiological studies have suggested a defect of the carnitine transport system in the plasma membrane in SCD patients5 and in the mouse model, juvenile visceral steatosis ( jvs; ref. 6 ). Although the responsible loci have been mapped in both human7 and mouse8, the underlying gene has not yet been identified. Recently, we cloned and analysed the function of a novel transporter protein termed OCTN2 ( ref. 9 ). Our observation that OCTN2 has the ability to transport carnitine in a sodium-dependent manner prompted us to search for mutations in the gene encoding OCTN2, SLC22A5 . Initially, we analysed the mouse gene and found a missense mutation in Slc22a5 in jvs mice. Biochemical analysis revealed that this mutation abrogates carnitine transport. Subsequent analysis of the human gene identified four mutations in three SCD pedigrees. Affected individuals in one family were homozygous for the deletion of a 113-bp region containing the start codon. In the second pedigree, the affected individual was shown to be a compound heterozygote for two mutations that cause a frameshift and a premature stop codon, respectively. In an affected individual belonging to a third family, we found a homozygous splice-site mutation also resulting in a premature stop codon. These mutations provide the first evidence that loss of OCTN2 function causes SCD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Treem, W.R., Stanley, C.A., Finegold, D.N., Hale, D.E. & Coates, P.M. Primary carnitine deficiency due to a failure of carnitine transport in kidney, muscle, and fibroblasts. N. Engl. J. Med. 17, 1331–1336 (1988).
Karpati, G. et al. The syndrome of systemic carnitine deficiency: clinical, morphologic, biochemical, and pathophysiologic features. Neurology 25, 16–24 (1975).
Roe, C.R. & Coates, P.M. Mitochondrial fatty acid oxidation disorder. in The Metabolic and Molecular Bases of Inherited Diseases (eds Scriver, C.R., Beaudet, A.L., Sly, W.S. & Valle, D.) 1508– 1509 (McGraw-Hill, New York, 1995).
Rinaldo, P., Stanley, C.A., Hsu, B.Y.L., Sabchez, L.A. & Stern, H.J. Sudden neonatal death in carnitine transporter deficiency. J. Pediatr. 131, 304–305 (1997).
Tein, I. et al. Impaired skin fibroblast carnitine uptake in primary systemic carnitine deficiency manifested by childhood carnitine-responsive cardiomyopathy. Pediatr. Res. 28, 247–255 (1990).
Hashimoto, N. et al. Gene-dose effect on carnitine transport activity in embryonic fibroblasts of JVS mice as a model of human carnitine transporter deficiency. Biochem. Pharmacol. 55, 1729– 1732 (1998).
Shoji, Y. et al. Evidence for linkage of human primary systemic carnitine deficiency with D5S436: a novel gene locus on chromosome 5q. Am. J. Hum. Genet . 63, 101–108 ( 1998).
Okita, K. et al. Definition of the locus responsible for systemic carnitine deficiency within a 1.6-cM region of mouse chromosome 11 by detailed linkage analysis. Genomics 33, 289–291 (1996).
Tamai, I. et al. Molecular and functional identification of sodium ion-dependent, high affinity human carnitine transporter OCTN2. J. Biol. Chem. 273, 20378–20382 ( 1998).
Walter, J.H. L-carnitine. Arch. Dis. Child. 74, 475– 478 (1996).
Bremer, J. Carnitine metabolism and functions. Physiol. Rev. 63 , 1420–1480 (1983).
Tripp, M.E. et al. Systemic carnitine deficiency presenting as familial endocardial fibroelastosis. A treatable cardiomyopathy. N. Engl. J. Med. 305, 385–390 (1981).
Chapoy, P.R. et al. Systemic carnitine deficiencya treatable inherited lipid-storage disease presenting as Reye's syndrome. N. Engl. J. Med. 303, 1389–1394 (1980).
Waber, L.J., Valle, D., Neill, C., DiMauro, S. & Shug, A. Carnitine deficiency presenting as familial cardiomyopathy: a treatable defect in carnitine transport. J. Pediatr. 101, 700–705 (1982).
Koizumi, T., Nikaido, H., Hayakawa, J., Nonomura, A. & Yoneda, T. Infantile disease with microvesticular fatty infiltration of viscera spontaneously occurring in C3H-H2-2 strain of mouse with similarities to Reye's syndrome. Lab Anim. 22, 83–87 (1988).
Horiuchi, M. et al. Primary defect of juvenile visceral steatosis (jvs) mouse with systemic carnitine deficiency is probably in renal carnitine transport system. Biochim. Biophys. Acta. 12, 25– 30 (1994).
Nikaido, H., Horiuchi, M., Hashimoto, N., Saheki, T. & Hayakawa, J. Mapping of jvs (juvenile visceral steatosis) gene, which causes systemic carnitine deficiency in mice, on chromosome 11. Mamm. Genome 6, 369– 370 (1995).
Tamai, I. et al. Cloning and characterization of a novel human pH-dependent organic cation transporter, OCTN1. FEBS Lett. 419, 107–111 (1997).
Matsuishi, T. et al. Successful carnitine treatment in two siblings having lipid storage myopathy with hypertrophic cardiomyopathy. Neuropediatrics 16, 6–12 (1985 ).
Claros, M.G. & von Heijne, G. TopPred II: an improved software for membrane protein structure predictions. Comput. Appl. Biosci. 10, 685–686 ( 1994).
Maiden, M.C., Davis, E.O., Baldwin, S.A., Moore, D.C. & Henderson, P.J. Mammalian and bacterial sugar transport proteins are homologous. Nature 325, 641–643 (1987).
Acknowledgements
We are grateful to the family members who participated in these studies. We also thank M.H. Jones, C. Schoenbach and H. Nomura for helpful advice, and colleagues in Group 1 for their technical assistance. This work was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Science, Sports and Culture, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nezu, Ji., Tamai, I., Oku, A. et al. Primary systemic carnitine deficiency is caused by mutations in a gene encoding sodium ion-dependent carnitine transporter. Nat Genet 21, 91–94 (1999). https://doi.org/10.1038/5030
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/5030
This article is cited by
-
A qualitative study on the perspectives of mothers who had been diagnosed with primary carnitine deficiency through newborn screening of their child
Orphanet Journal of Rare Diseases (2023)
-
Embryonic exposure to acetyl-L-carnitine protects against valproic acid-induced cardiac malformation in zebrafish model
Amino Acids (2023)
-
High-fat diet-induced nonalcoholic steatohepatitis is accelerated by low carnitine and impaired glucose tolerance in novel murine models
Laboratory Investigation (2022)
-
Gut microbiota production of trimethyl-5-aminovaleric acid reduces fatty acid oxidation and accelerates cardiac hypertrophy
Nature Communications (2022)
-
Ergothioneine and central nervous system diseases
Neurochemical Research (2022)