Abstract
Despite the paradigm that the innate immune system uses nucleic acid–specific receptors to detect viruses because of a lack of other conserved features, many viruses are recognized by Toll-like receptor 2 (TLR2) and TLR4. The relevance of this recognition for antiviral immunity remains largely unexplained. Here we report that TLR2 activation by viruses led to the production of type I interferon. TLR2-dependent induction of type I interferon occurred only in response to viral ligands, which indicates that TLR2 is able to discriminate between pathogen classes. We demonstrate that this specialized response was mediated by Ly6Chi inflammatory monocytes. Thus, the innate immune system can detect certain non–nucleic acid features of viruses and links this recognition to the induction of specific antiviral genes.
Similar content being viewed by others
Main
Receptors of the innate immune system have evolved to recognize conserved microbial features that represent broad pathogen classes1. This strategy ensures that diverse pathogen species can be quickly recognized by the host, as long as these microbial features are sufficiently constrained so that they remain invariant. Examples of such features are the bacterial cell wall components lipopolysaccharide and peptidoglycan. Members of the Toll-like receptor (TLR) family recognize those and other microbial ligands and induce signals important for initiation of both innate and adaptive immunity1. Thus, mice lacking TLR function show greater susceptibility to infection.
Viral recognition by the innate immune system is more challenging than recognition of other pathogen classes because of the relative paucity of conserved features2. Viruses replicate in host cells, and they do not generate any of the unique biochemical products present in bacterial and fungal cell walls. It has been argued that this lack of conserved viral features has forced the innate immune system to use nucleic acid as a means of detecting viral infection. Indeed, several members of the TLR family recognize nucleic acids: TLR3 recognizes double-stranded RNA; TLR7 and TLR8 recognize single-stranded RNA; and TLR9 recognizes CpG motifs in DNA2. In addition, a family of cytosolic receptors, including RIG-I, Mda5, DAI and AIM2, recognizes various nucleic acid species in the cytosol2,3,4,5,6. Targeting nucleic acids allows the recognition of highly diverse viral species with only a few innate receptors.
One of the key components of antiviral immunity is induction of the type I interferon family of cytokines (called 'type I interferon' here)7. Type I interferon induces hundreds of genes that promote an antiviral state in cells. The importance of this signaling network is illustrated by the extreme susceptibility to viral infection of mice lacking the type I interferon receptor8. All of the nucleic acid–sensing TLRs induce type I interferon, which emphasizes the importance of this cytokine family in antiviral immunity. For TLR7 and TLR9, however, induction of type I interferon only occurs in plasmacytoid dendritic cells (pDCs) via the common signaling adaptor MyD88. In other cell types, activation of TLR7 and TLR9 does not lead to the production of type I interferon9. Similarly, most TLRs involved in bacterial or fungal recognition (TLR2 and TLR5) are not expressed in pDCs10 and do not induce type I interferon in other cell types. The notable exception is TLR4, which can induce type I interferon in macrophages and conventional DCs via the signaling adaptor TRIF11. Nevertheless, type I interferon is clearly less critical for antibacterial immunity than for antiviral immunity12,13.
By many criteria, viral proteins would seem poor choices as targets for innate receptors compared with nucleic acids. First, any given viral protein is unlikely to be shared among diverse viruses. Second, innate recognition of a viral protein would probably select for mutants that escape recognition yet retain function, if at all possible. Nevertheless, several viruses do encode proteins that are able to stimulate TLR2, a receptor known to recognize multiple bacterial and fungal cell wall components. The best-characterized example is stimulation of TLR2 by glycoprotein B from human cytomegalovirus14,15, but mouse cytomegalovirus (MCMV)16, herpes simplex virus types 1 and 2 (refs. 17,18), hepatitis C virus19, lymphocytic choriomeningitis virus20, measles virus21 and vaccinia virus22 are also able to stimulate TLR2. In some of these cases, it seems that viruses benefit in some way from stimulating TLRs. For example, measles virus may have evolved the ability to activate TLR2 as a means of upregulating the viral entry receptor CD150 (ref. 21). In other examples, however, there is evidence that TLR2 activation contributes to protection. Most notably, mice lacking TLR2 are impaired in their ability to mount an innate or adaptive immune response to vaccinia virus22. One problematic aspect of any general role for TLR2 in antiviral immunity, however, is the apparent inability of this receptor to induce type I interferon23,24,25.
Here we describe a specialized role for TLR2 in the innate recognition of several viruses. In contrast to the well-documented transcriptional response induced by bacterial ligands, we show that TLR2 induced type I interferon when activated by viruses. This signaling pathway was unique to 'inflammatory' monocytes (IMs). The functional specialization of these cells is conceptually analogous to the role of pDCs in TLR7 and TLR9 signaling and probably represents a general strategy for achieving specificity in innate immune signaling.
Note: Supplementary information is available on the Nature Immunology website.
Results
TLR2-mediated recognition of DNA viruses
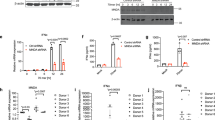
TLR2 has been linked to the recognition of several DNA viruses, including vaccinia virus, human cytomegalovirus, MCMV, and herpes simplex virus types 1 and 2. These viruses contain ligands that can activate additional TLRs (for example, TLR9), so we first sought to determine the relative contribution made by TLR2 for viral recognition. To minimize potential viral interference with innate immune signaling, we inactivated each virus with ultraviolet irradiation before stimulating cells. Both vaccinia virus and MCMV induced activation of the transcription factor NF-κB in HEK293 human epithelial cells stably expressing mouse TLR2 but not in control cells lacking TLR2 (Fig. 1a), which supports the idea that TLR2 is involved in the recognition of these viruses. We directly tested the relative contributions of TLR2 and TLR9 by stimulating bone marrow–derived dendritic cells (DCs) from TLR2-deficient, TLR9-deficient, adaptor protein Myd88–deficient and control mice. Unexpectedly, we found that the production of tumor necrosis factor (TNF) by DCs was entirely TLR2 dependent (Fig. 1b). TLR9 seemed to have little role in these cells. We obtained similar results with bone marrow–derived macrophages (Fig. 1c). These data suggest that TLR2 may have a chief role in the recognition of certain viruses.
(a) Luciferase activity in HEK293 cells stably transfected with a NF-κB luciferase reporter (HEK293) or the reporter together with plasmids encoding human TLR2 and human CD14 (HEK293-TLR2), then left unstimulated (Unstim) or stimulated with ultraviolet irradiation–inactivated MCMV or vaccinia virus (VV; multiplicity of infection (MOI), horizontal axes) and assessed 10 h after activation. Pam3SK4 (Pam3) serves as a positive control. Results are presented relative to activation in unstimulated cells. (b,c) Flow cytometry of intracellular TNF in bone marrow–derived DCs (BMDC; b) or macrophages (MΦ; c) stimulated with TLR ligands or viruses (above plots). Plots of stimulated cells (black lines) are overlaid on plots of unstimulated cells (shaded). DC plots (b) are gated on CD11c+ cells. Genotype (right margin): WT, wild-type; KO, knockout. Data are representative of at least three experiments (error bars (a), s.d.).
TLR2 induces type I interferon
The lack of TLR9 activation in DCs and macrophages treated with vaccinia virus or MCMV is at odds with the well-documented role of TLR9 in the recognition of DNA viruses26. One potential explanation for this discrepancy is that TLR9-mediated recognition of virus is best observed in pDCs. To address this possibility, we used bone marrow cells as a source of ex vivo pDCs and measured the production of type I interferon in response to MCMV and vaccinia virus. As expected, MCMV induced potent production of type I interferon and this response was lower in TLR9-deficient cells (Fig. 2a). Unexpectedly, the production of type I interferon in response to MCMV was also partially TLR2 dependent. Moreover, the production of type I interferon in response to vaccinia virus was entirely TLR2 dependent (Fig. 2a). We found a similar dependence on TLR2 in splenocytes (data not shown).
(a–c) Bioassay of type I interferon in supernatants of bone marrow cells (genotype, key) 24 h after stimulation (horizontal axis). B6, C57BL/6; Unc93b1, 3d. (d) Real-time PCR analysis of transcripts for IFN-β and IFN-α4 in bone marrow cells stimulated with vaccinia virus or CpG oligonucleotides, presented relative to expression of mRNA for the ribosomal protein S17 (RPS17). Data are representative of at least two experiments (error bars, s.d.).
There is no known pathway by which TLR2 can induce type I interferon, so a requirement for TLR2 in interferon production by MCMV and vaccinia virus was unexpected. There are two known mechanisms for the induction of type I interferon by TLR family members. TLR3 and TLR4 induce activation of the transcription factor IRF3 via the signaling adaptor TRIF11. TLR7 and TLR9 can induce IRF7 activation 'downstream' of MyD88, but this interaction occurs only in pDCs, which lack TLR2 expression10. To define the signaling pathway responsible for TLR2-dependent production of type I interferon, we examined the requirement for several known TLR signaling components. The production of type I interferon in response to vaccinia virus or MCMV was much lower in MyD88-deficient cells and cells doubly deficient in MyD88 and TRIF but not in TRIF-deficient cells (Fig. 2a). Both IRF3 and IRF7 seemed to contribute to the production of type I interferon induced by TLR2 because cells doubly deficient in IRF3 and IRF7 were completely impaired in the production of type I interferon (Fig. 2b). In contrast, the response to vaccinia virus was unaffected in IRF1-deficient cells (Supplementary Fig. 1a). As expected, production of type I interferon was also impaired in cells lacking the type I interferon receptor (Supplementary Fig. 1b). To rule out the possibility of any contribution by the nucleic acid–sensing TLRs, we tested the response to vaccinia virus in 3d mice, which have a nonfunctional allele of Unc93b1. These mice respond normally to TLR2 and TLR4 ligands but are unable to respond to TLR3, TLR7 and TLR9 ligands27. Notably, the response to vaccinia virus in cells from 3d mice was equivalent to that of wild-type control mice (Fig. 2c), which ruled out the possibility of involvement of nucleic acid recognition in the production of type I interferon. Finally, to address whether RNA polymerase III–dependent activation of RIG-I or Mda5 was involved in the induction of type I interferon28,29, we examined the response of cells lacking the common signaling adaptor MAVS. The production of type I interferon in response to vaccinia virus was equivalent in MAVS-deficient and control cells (Supplementary Fig. 1c), which ruled out the possibility of involvement of this pathway. Collectively these data indicate that in response to virus, a MyD88-dependent pathway 'downstream' of TLR2 leads to activation of IRF3 and IRF7 and the transcription of genes encoding type I interferon.
We next measured which specific genes encoding type I interferon were induced by TLR2 in response to viruses. We focused on the vaccinia virus response because unlike MCMV, recognition of vaccinia virus was entirely TLR2 dependent. Vaccinia virus induced the production of interferon-β (IFN-β) and IFN-α4 by bone marrow cells in a TLR2-dependent manner (Fig. 2d). The induction of each cytokine peaked at 12 h, whereas the response to CpG oligonucleotides peaked at 4 h. It is known that pDCs are responsible for the rapid type I interferon induction in response to CpG oligodeoxynucleotide30. The delayed response to vaccinia virus suggested that a cell type other than pDCs may have been responsible for the TLR2 response.
Differences in TLR2 responses to viral and bacterial ligands
The data presented thus far contradict a large body of work demonstrating that TLR2 does not induce type I interferon23,24,25. One major difference between our work here and previously published studies is the use of viral rather than bacterial TLR2 ligands. To address whether the nature of the microbial ligands could account for differences in the induction of type I interferon, we compared the induction of IFN-β and IFN-α4 in response to vaccinia virus and TLR2 ligands. In contrast to vaccinia virus, the triacylated lipid Pam3SK4 (a TLR2-TLR1 agonist31) did not induce IFN-β or IFN-α4 in bone marrow cultures (Fig. 3a).
(a) IFN-β transcripts in bone marrow cells left unstimulated or treated for 12 h with Pam3SK4 or vaccinia virus, presented relative to the expression of RPS17 mRNA. (b) Flow cytometry of YFP production by bone marrow cells (BM) or splenocytes (Spleen) obtained from MOB mice and left unstimulated or stimulated for 20 h with Pam3SK4, FSL-1 or vaccinia virus. Numbers in bottom right corners indicate percent YFP+ cells. Data are representative of at least three experiments (error bars (a), s.d.).
As an alternative way of measuring IFN-β induction, we used IFN-β reporter mice in which cDNA encoding yellow fluorescent protein (YFP) has been 'knocked in' downstream of the IFN-β locus (MOB mice)32. YFP was detectable in bone marrow and splenocytes from these mice after stimulation with vaccinia virus. In contrast, there was no YFP signal when the same cells were stimulated with Pam3SK4 or FSL-1 (a TLR2-TLR6 agonist33; Fig. 3b). This lack of production of type I interferon was not due to poor stimulation because TNF was induced by all TLR2 ligands (Supplementary Fig. 2). Thus, a population of cells in the bone marrow and spleen is able to discriminate between viral and bacterial TLR2 stimuli and selectively induce type I interferon in response to virus.
Both bone marrow–derived DCs and macrophages express TLR2 and respond to vaccinia virus. If the ability to discriminate between bacterial and viral ligands is a general property of TLR2 when it is expressed on all cells, then TLR2-dependent production of type I interferon would be expected in response to viral ligands. However, the production of type I interferon by bone marrow–derived DCs and HEK293T cells was TLR2 independent and was probably due to activation of the cytosolic DNA sensor(s) (Supplementary Fig. 3). Therefore, the ability to produce type I interferon in a TLR2-dependent manner is probably restricted to a specialized cell type present in spleen and bone marrow.
IMs produce TLR2-dependent type I interferon
We next sought to identify the population of cells in the bone marrow and spleen responsible for TLR2-dependent induction of type I interferon. As an initial approach, we used magnetic bead cell sorting to separate bone marrow cells on the basis of their expression of the common myeloid marker CD11b or the common DC marker CD11c. Notably, sorted CD11b+ cells were enriched for TLR2-dependent production of type I interferon in response to vaccinia virus, whereas the CD11b− cells no longer responded (Fig. 4a). In contrast, sorting on the basis of CD11c did not enrich for cells producing interferon (Fig. 4b). We obtained similar results with cells from transgenic mice expressing the diphtheria toxin receptor (DTR) driven by the promoter of the gene encoding CD11b (CD11b-DTR mice)34. Splenocytes from CD11b-DTR mice injected with diphtheria toxin no longer responded to vaccinia virus (Fig. 4c). These results suggest that a CD11b+CD11c− population of cells was responsible for the TLR2-dependent production of type I interferon.
(a,b) Bioassay of type I interferon in supernatants of bone marrow cells collected from B6 or TLR2-deficient mice and sorted into CD11b+ and CD11b− populations (a) or CD11c+ and CD11c− populations (b), then left unstimulated or stimulated for 24 h with CpG or vaccinia virus. (c) Bioassay of type I interferon in supernatants of splenocytes collected from CD11b-DTR–transgenic mice 24 h after injection with saline (PBS) or diphtheria toxin (DT), then left unstimulated or stimulated for 24 h with CpG or vaccinia virus. Data are representative of at least three experiments (error bars, s.d.).
Although the lack of CD11c expression suggested that pDCs were not responsible for the TLR2-dependent production of type I interferon, we addressed this possibility directly with MOB mice. We compared the surface markers expressed by responding (YFP+) cells in bone marrow and spleen stimulated with vaccinia virus or CpG oligonucleotides. CpG oligonucleotides induced TLR9-dependent production of type I interferon by pDCs, and the YFP+ cells in these cultures were B220+CD11c+, a surface phenotype consistent with that of pDCs (Fig. 5a). In contrast, the cells that responded to vaccinia virus were B220− and had lower expression of CD11c (Fig. 5a). These distinct surface phenotypes demonstrate that different cell types were responding to vaccinia virus and CpG oligonucleotides.
(a,b) Flow cytometry of splenocytes or bone marrow cells collected from MOB mice, stimulated for 20 h with CpG or vaccinia virus and stained with antibody to B220 (anti-B220) or anti-CD11c (a) or with anti-Ly6C, anti-CD11b or anti-Ly6G (b). Orange lines, YFP-gated cells; shaded histograms, total ungated cells. (c) Flow cytometry of bone marrow cells and splenocytes stained with anti-CD11b, anti-Ly6C and anti-TLR2. (d) Frequency of YFP+ cells among bone marrow cells obtained from MOB mice and cultured in the presence (mAb to TLR2) or absence (No mAb) of a TLR2-blocking monoclonal antibody, then left unstimulated or stimulated with Pam3SK4, CpG or vaccinia virus. (e) Flow cytometry of bone marrow cells sorted on the basis of expression of CD11b and Ly6C (left) and postsort analysis (middle) of populations within (positive (+IM)) or outside (negative (−IM)) the Ly6ChiCD11b+CD11c−B220− gate. B220+ and CD11c+ cells were excluded by gating; numbers above outlined areas indicate percent IMs. Right, bioassay of the production of type I interferon by positive (+IM) and negative (−IM) populations left unstimulated or stimulated for 24 h with CpG A or vaccinia virus. Data are representative of at least three experiments (error bars (e), s.d.).
To characterize more completely the CD11b+ cells producing IFN-β in response to vaccinia virus, we stained bone marrow cells and splenocytes with a panel of antibodies to common hematopoietic surface markers. On the basis of the absence of certain lineage markers, we were able to exclude B cells, T cells, natural killer cells and neutrophils as the source of type I interferon (data not shown). The expression of CD11b suggested that the cells responding to vaccinia virus may represent a subset of monocytes. Monocytic subsets have been classified on the basis of differences in expression of the surface markers Ly6C and Ly6G35,36,37. Cells expressing YFP in response to vaccinia virus were Ly6ChiLy6G− (Fig. 5b), which suggested that they represent the subset of monocytes often referred to as 'inflammatory' monocytes. Although the expression of CD11c on IMs is inconsistent with published reports describing these cells, one possible explanation for this discrepancy is that activation of these cells leads to CD11c upregulation35,37. Given that and the finding that sorting based on CD11c staining did not alter the response to vaccinia (Fig. 4b), we conclude that the cells that make TLR2-dependent type I interferon are not initially CD11c+, but upregulate CD11c expression after stimulation with virus. Staining with a TLR2-specific antibody confirmed that IMs in the spleen and bone marrow expressed TLR2 (Fig. 5c). Moreover, treatment of MOB bone marrow with TLR2-blocking antibodies before stimulation with vaccinia virus resulted in a much lower number of YFP+ cells (Fig. 5d), which demonstrates that the production of type I interferon by these cells requires TLR2.
To demonstrate formally that IMs were solely responsible for the TLR2-dependent type I interferon, we sorted these cells on the basis of expression of Ly6C, CD11b, CD11c, and B220 (Fig. 5e). Ly6ChiCD11b+CD11c−B220− cells sorted from the bone marrow produced type I interferon when stimulated with vaccinia virus in a TLR2-dependent manner. Moreover, the 'negative' population (that is, all other cells outside the Ly6ChiCD11b+CD11c−B220− gate) did not produce type I interferon in response to virus (Fig. 5e), which indicated that IMs were solely responsible for the response. Notably, these 'negative' cells did produce type I interferon when stimulated with CpG oligonucleotides, whereas the IMs did not, which demonstrated that our sorting parameters had effectively separated pDCs from IMs. Finally, quantitative PCR analysis of cDNA generated from the sorted IMs confirmed that they had higher expression of the chemokine receptor CCR2, as previously reported for these cells36, and we observed slightly higher expression of IRF7 transcripts in IMs, although not as high as the expression in pDCs (Supplementary Fig. 4). These data suggest that Ly6Chi IMs have the unique ability to link viral recognition by TLR2 to the production of type I interferon.
Role of IMs during viral infection
Our in vitro analyses of cells from bone marrow and spleen linked IMs to the recognition of vaccinia virus and suggested that TLR2 activation in these cells led to the production of type I interferon. To address the relevance of these cells during vaccinia virus infection in vivo, we used the CD11b-DTR mice described above. Although the DTR transgene is driven by the promoter of the gene encoding CD11b, previous analyses of these mice have demonstrated that only a limited population of CD11b+ cells are deleted after injection of diphtheria toxin34. Although monocytes and some tissue-resident macrophages are removed, other CD11b+ cells (such as neutrophils and activated lymphocytes) remain largely unaffected. Indeed, we observed similar numbers of Ly6G+CD11b+ neutrophils in mice injected with diphtheria toxin or PBS, and the overall profile of CD11b-expressing cells in the spleen remained mostly unchanged (Fig. 6a). In contrast, IMs were deleted efficiently, thus providing a system with which to probe the functional relevance of these cells in vivo. Notably, when we subsequently challenged DTR-injected mice with vaccinia virus, serum concentrations of type I interferon were diminished to amounts similar to those in uninfected mice (Fig. 6b). Injection of diphtheria toxin into nontransgenic mice followed by vaccinia virus infection had no effect on the production of type I interferon, as expected (Supplementary Fig. 5). To assess the relevance of these cells to viral clearance, we measured viral titers in CD11b-DTR mice depleted of IMs before infection. Mice lacking IMs had higher titers of vaccinia virus in the liver and ovaries (Fig. 6c and Supplementary Fig. 6). Collectively these data indicate that IMs are a key early source of type I interferon during viral infection and are necessary for early restriction of viral replication.
(a) Flow cytometry of splenocytes collected from CD11b-DTR–transgenic mice 24 h after intravenous injection of PBS or diphtheria toxin, then stained with anti-Ly6C, anti-CD11b or anti-Ly6G. Numbers adjacent to outlined areas indicate Ly6G+CD11b+ cells (top row) or Ly6C+CD11b+ cells (bottom row).(b) Bioassay of type I interferon in serum from CD11b-DTR–transgenic mice injected with PBS or diphtheria toxin 24 h before infection with vaccinia virus (1 × 106 plaque-forming units), assessed 24 h after infection. (c) Viral titers in the liver of CD11b-DTR–transgenic mice treated as described in b, assessed 48 h after infection. Each symbol represents an individual mouse; small horizontal lines indicate the mean. P value, unpaired t-test. Data are representative of at least two experiments.
Interferon induction requires TLR2 internalization
Finally, we sought to address the selective production of type I interferon in response to viruses by IMs. Because IMs produce TNF in response to both viral and bacterial ligands, the difference in type I interferon responses was unlikely to be due to lack of recognition. Studies of TLR4 signal transduction have shown that induction of type I interferon requires receptor internalization, whereas signals that lead to the production of TNF and interleukin 6 can occur at the plasma membrane38. To determine whether similar cell biological regulation controls the production of type I interferon 'downstream' of TLR2, we blocked endocytosis with the actin-depolymerizing drug cytochalasin D or blocked endosomal maturation with chloroquine. Treatment of bone marrow cultures with either inhibitor before stimulation with vaccinia virus completely abrogated the production of type I interferon (Fig. 7a). In contrast, the production of TNF was unaffected (Fig. 7b), which indicates that these agents do not prevent overall recognition of vaccinia virus or signaling by TLR2. Instead, receptor internalization seems necessary only for TLR2-dependent production of type I interferon.
(a) Flow cytometry of YFP+ cells among bone marrow obtained from MOB mice and then incubated with 15 μM chloroquine, 1 μM cytochalasin D or left untreated before stimulation for 20 h with vaccinia virus or CpG. (b) Flow cytometry of intracellular TNF in IMs treated as described in a, measured by intracellular cytokine staining. Data are representative of at least two experiments.
Discussion
Here we have reported the identification of a TLR2-dependent antiviral signaling pathway that leads to the production of type I interferon. Before this work, TLR2-dependent production of type I interferon had not been reported to our knowledge, and indeed in most cell types, TLR2 does not induce this antiviral response. We have demonstrated that IMs were uniquely able to respond to viral TLR2 ligands by producing type I interferon, and our work suggests that these cells represent another specialized antiviral cell type with functional and conceptual parallels to pDCs. In addition, our data solidify the interpretation that viral recognition by TLR2 is a host strategy, rather than a manipulation by viruses, and indicate that certain viral proteins are sufficiently constrained to serve as targets for innate immune receptors. Overall, our work has implications for understanding how the innate immune system recognizes viruses.
Until recently, the specific and differing roles of monocytic subpopulations during immune responses have not been well appreciated. However, several studies have identified IMs as a largely bone marrow–resident cell type that is rapidly recruited to sites of infection in a CCR2-dependent manner35,36,37. These cells have been called 'inflammatory' monocytes to distinguish them from Ly6C− monocytes, which are thought to be more important in maintaining tissue homeostasis35. IMs can differentiate into many different DC subsets at sites of inflammation, including 'TipDCs' (DCs that produce TNF and inducible nitric oxide synthase) as well as inflammatory DCs35,36,37. These cells have been linked to bacterial, parasitic and viral immunity37. An additional role for Ly6Chi monocytes has been reported in a mouse model of induced lupus. In this model, Ly6Chi monocytes accumulate in the peritoneal cavity of mice after injection of 2,6,10,14-tetramethylpentadecane39. Unexpectedly, the Ly6Chi monocytes express type I interferon in this model. Although the activation signal for these cells has not been defined in this context, the observation that IMs can produce type I interferon during disease supports our contention that they may function as specialized interferon-producing cells. The ability of IMs to secrete proinflammatory cytokines and type I interferon suggests that these cells may have a key role early during viral infection. In addition, the differentiation of IMs into DCs after activation by viruses may further enhance their contribution to the antiviral immune response through the induction of adaptive responses.
An unexpected aspect of the TLR2 function on IMs is the ability to distinguish between viral and bacterial ligands; type I interferon is induced only in response to viral ligands, whereas TNF production occurs in response to both classes of stimuli. This difference in response may be partially explained by the observation that activation of the signal-transduction pathway leading to the production of type I interferon requires receptor internalization, as both chloroquine and cytochalasin D disrupted interferon production but not TNF production. This dichotomy is reminiscent of TLR4 signaling, in which TRIF activation occurs at endosomal membranes, whereas MyD88 activation occurs at the plasma membrane38. Our data suggest that TLR2 signaling is also regulated through localization, yet in contrast to TLR4 signaling, all TLR2 signaling is MyD88 dependent. In this sense, differences in signaling by TLR2 may be more conceptually similar to TLR9 signaling in that different ligands produce distinct responses through the same signaling adaptor (MyD88). For example, in pDCs, class B CpG oligonucleotides lead to the production of TNF and interleukin 12 but not to the production of type I interferon, whereas class C CpG oligonucleotides lead to the production of TNF, interleukin 12 and type I interferon9,40. IMs have a similar ability to use one TLR and produce two unique responses. How such specificity can be generated 'downstream' of a particular TLR is not understood in pDCs or in our system. It is possible that viruses may be more efficiently internalized or trafficked to a specialized compartment from which type I interferon signaling can be initiated. Still, such a mechanism cannot explain why conventional DCs or macrophages are unable to make type I interferon in a TLR2-dependent manner when stimulated with virus. Thus, it seems possible that a specialized coreceptor might be needed to generate the specificity that we have identified.
The finding that innate immune recognition of vaccinia virus is so heavily TLR2 dependent is somewhat unexpected given the additional innate receptors capable of viral recognition. For example, TLR9 is clearly involved in the innate recognition of many DNA viruses, yet our data as well as the work of others22 suggest that TLR9 does not have a major role in the recognition of vaccinia virus. The ability of some viruses to evade certain innate receptors has undoubtedly required the host to evolve additional strategies of viral recognition. Cytosolic DNA sensors are important in the detection of viral nucleic acid in the host cytosol41,42,43. In DCs and macrophages stimulated with vaccinia virus, we found a TLR-independent type I interferon response, which was presumably due to activation of cytosolic DNA sensors. In IMs as well as during in vivo infection, however, induction of type I interferon is largely TLR2 dependent. This dependence suggests that vaccinia virus can evade innate receptors, such as TLR9 and cytosolic DNA sensors, yet remains detectable by TLR2.
Although nucleic acid recognition seems to be the dominant strategy used by the innate immune system to detect viral infection, there is accumulating evidence that the host can recognize certain viruses independently of nucleic acid. The work presented here supports this view. Our data and the work of others suggest that the innate immune system must have evolved specificity for certain viral proteins that are unable to mutate and escape recognition. Such a scenario is not unprecedented. Bacterial flagellin is recognized by several innate receptors (TLR5 (ref. 44), Ipaf45,46 and Naip5 (ref. 47)), yet most bacteria seem unable to mutate flagellin to avoid recognition. Importantly, mutations that abrogate flagellin recognition result in nonmotile bacteria47,48. Thus, it would seem that certain pathogen-encoded proteins are sufficiently constrained that they can serve as targets for innate immune receptors.
According to that reasoning, it is possible that the innate immune system has evolved to recognize viral proteins that are under functional constraints similar to those of flagellin. The viral fusion apparatus is a particularly likely target in this context. All viruses have a fusion apparatus that is absolutely essential for propagation, which makes it an ideal target for the innate immune system. Although fusion proteins from unrelated viruses share no homology at the amino acid level, structural studies have demonstrated that in some cases, these proteins share unexpected structural homology49. It is possible that TLR2 has evolved to recognize a conserved structural feature associated with certain viral fusion proteins. In support of that hypothesis, human cytomegalovirus glycoprotein B, which is required for viral fusion, has been reported to activate TLR2 (ref. 14). Additionally, many studies suggest that TLR4 can recognize viral structural proteins50,51,52,53,54. The identification of ligands from other viruses recognized by TLR2 and TLR4 will be necessary to address whether a common structural feature is targeted.
Methods
Mice and viruses.
C57BL/6, FVB/N, FVB/N-CD11b-DTR and Irf1−/− mice were from Jackson Laboratories. UNC93b1-mutant mice were from the Mutant Mouse Regional Resource Centers. Tlr2−/−, Tlr9−/−, Myd88−/− and Myd88−/−Trif−/− mice (provided by S. Akira) were backcrossed at least seven generations onto the C57BL/6 background. Irf3−/− and Ifnar1−/− (IFN-αβ receptor–deficient) mice were provided by D. Portnoy; Irf7−/− and Irf3−/−Irf7−/− mice were provided by K. Fitzgerald; and Mavs−/− mice were provided by Z. Chen. MOB mice have been described32. All mice were housed in the animal facilities at the University of California, Berkeley according to guidelines of the Institutional Animal Care and Use Committee.
Vaccinia virus (Western Reserve strain) was from D. Raulet; MCMV (Smith strain) was from L. Coscoy. Vaccinia virus was propagated on and titers were quantified by plaque assay on BHK21 hamster kidney cells. MCMV was propagated on and titers were quantified by plaque assay on NIH3T3 fibroblast cells.
Flow cytometry and fluorescence-activated cell sorting.
The following antibodies were from eBioscience or BD Biosciences: allophycocyanin-indotricarbocyanine– or phycoerythrin-indodicarbocyanine –conjugated anti-CD11b (M1/70); fluorescein isothiocyanate– or phycoerythrin-indotricarbocyanine–conjugated anti-CD11c (N418); phycoerythrin-indodicarbocyanine– or fluorescein isothiocyanate–conjugated anti-B220 (RA3-6B2); peridinin chlorophyll protein–cyanine 5.5–conjugated anti-Ly6C (HK1.4); phycoerythrin- or fluorescein isothiocyanate–conjugated anti-Ly6G (1A8); phycoerythrin- or Pacific blue–conjugated anti-TNF (MP6-XT22); and phycoerythrin-conjugated anti-TLR2 (T2.5). Before being stained for surface markers, cells were incubated with anti-CD16-CD32 (2.4G2; Monoclonal Antibody Core, University of California, San Francisco). Cells were stained for intracellular cytokines with a Fixation & Permeabilization kit according to manufacturer's instructions (eBioscience). Brefeldin was added 30 min after stimulation and cells were collected after an additional 5 h. Data were collected on an LSR II (Becton Dickinson) or FC-500 (Beckman Coulter) and were analyzed with FloJo software (TreeStar).
Cells were sorting on an Influx cell sorter (Becton Dickinson). IMs were isolated as described above. Sorted pDCs (Supplementary Fig. 4) were purified from bone marrow by sorting of B220+Cd11c+ cells.
Cell lines and tissue culture.
HEK293, NIH3T3 and BHK21 cells were from American Type Culture Collection; HEK293-TLR2 cells have been described17. ISRE-L929 cells were from D. Portnoy55 and have been described: these are L929 mouse fibroblast cells stably transfected with an interferon-sensitive responsive element upstream of the luciferase gene.
Cell lines were cultured in DMEM supplemented with 10% (vol/vol) FCS, L-glutamine, penicillin-streptomycin, sodium pyruvate and HEPES, pH 7.2 (Invitrogen). Primary cells were cultured in RPMI-1640 medium supplemented with 10% (vol/vol) FCS, L-glutamine, penicillin-streptomycin, sodium pyruvate and HEPES, pH 7.2.
Bone marrow–derived conventional DCs and macrophages were differentiated in RPMI medium supplemented with supernatant containing granulocyte-macrophage colony-stimulating factor or granulocyte-macrophage colony-stimulating factor, respectively, as described56. After 5 d, cells were replated in RPMI media and stimulated.
Luciferase assay.
ISRE-L929 cells were used to measure type I interferon in supernatants or serum. In parallel, ISRE-L929 cells were treated with serial dilutions of recombinant IFN-β (R&D Systems) to generate a standard curve in each experiment. In some cases, supernatants or serum were diluted to achieve signal in the linear range of the standard curve.
All cells were lysed with Passive Lysis buffer (Promega) and luciferase was measured on an LMax II384 luminometer (Molecular Devices).
Collection and stimulation of bone marrow and spleen cells.
Spleens were dispersed into single-cell suspensions by passage through nylon mesh (BD Falcon). Bones were washed with ethanol and RPMI media, and bone marrow cells were released by gentle grinding with a mortar and pestle. Cells were passed through a filter for removal of debris, red blood cells were removed by hypotonic lysis and the remaining cells were washed three times with RPMI media, counted and plated.
All viral stimulations were done at an MOI of 0.2 with particles inactivated by ultraviolet irradiation, unless otherwise noted. CpG oligonucleotide (5′-TCCATGACGTTCCTGACGTT-3′, with phosphorothioate linkages; IDT or Invitrogen) and CpG A oligonucleotide (5′-G*GTGCATCGATGCAG*G*G*G*G*G-3′ (asterisks indicate phosphorothioate linkages); IDT) were each used at a concentration of 1 μM. Cultures were stimulated with FSL-1, Pam3SK4 or resiquimod (TLR8 agonist R-848; Invivogen) at a concentration of 0.1 μg/ml, 1 μg/ml or 1 μg/ml, respectively. TLR2-blocking antibody (T2.5; eBioscience) was added to cells at a concentration of 50 μg/ml 30 min before stimulation.
Quantitative real-time RT-PCR.
For RT-PCR analysis of IFN-β and IFN-α4, cells were collected at various times after stimulation and resuspended in TRIzol. For RT-PCR analysis of CCR2 and IRF7, cells were sorted on a BD Influx and were resuspended in TRIzol.
For all samples, the TRIzol Plus RNA Purification system (Invitrogen) was used for isolation of RNA. An ABI7300 Real-Time PCR system (Applied Biosystems) with SYBR Green–based quantification was used for quantitative PCR. Gene-specific transcript amounts were normalized to the amount of RPS17 mRNA. The following primers were used: RPS17, 5′-CGCCATTATCCCCAGCAAG-3′ and 5′-TGTCGGGATCCACCTCAATG-3′; IFN-β, 5′-ATAAGCAGCTCCAGCTCCAA-3′ and 5′-CTGTCTGCTGGTGGAGTTCA-3′; IFN-α4, 5′-GCAATGACCTCCATCAGCAGC-3′ and 5′-CACTCCTTCTCCTCACTCAGTCTTG-3′; IRF7, 5′-CTACACCATCTACCTGGGTTTTGG-3′ and 5′-AGACAAGCACAAGCCGAGACTG-3′; and CCR2, 5′-TCCTTGGGAATGAGTAACTGTGT-3′ and 5′-TGGAGAGATACCTTCGGAACTT-3′.
Magnetic cell sorting.
CD11b+ and CD11c+ bone marrow cells were positively sorted by magnetic cell sorting. Cells were stained with biotinylated anti-CD11b or anti-CD11c then were incubated with anti-biotin microbeads (Miltenyi Biotec). Cells were sorted on an AutoMACS cell sorter (Miltenyi Biotec). The positive and negative (flow-through) populations were collected. Positive populations were enriched for target cells but also contained CD11b− or CD11c− cells.
CD11b-DTR mice.
CD11b-DTR mice were injected intravenously with 500 ng diphtheria toxin (Sigma). For in vitro experiments, splenocytes were collected 24 h later and stimulated with vaccinia virus (MOI, 0.2) or CpG oligonucleotide (1 μM). Type I interferon was measured by bioassay in supernatants after 24 h.
For in vivo experiments, mice were infected with live vaccinia virus (1 × 106 plaque-forming units) 24 h after injection of diphtheria toxin. At 24 h after infection, serum was collected and type I interferon was measured by bioassay. At 48 h after infection, organs were collected and plaque-forming units were counted.
Inhibition of receptor internalization.
For measurement of interferon production, bone marrow from MOB mice was treated with chloroquine (15 μM) or cytochalasin D (1 μg/ml) for 30 min before the addition of ligands. At 20 h after stimulation, cells were collected and analyzed.
For measurement of TNF production, bone marrow from C57BL/6 mice was pretreated with chloroquine (15 μM) or cytochalasin D (1 μM) for 14 h before the addition of ligands. At 30 min after the addition of ligands, brefeldin was added. Cells were collected after an additional 5 h.
Change history
12 November 2009
In the version of this article initially published, the label along the vertical axis of the top row in Figure 6a is incorrect. The correct label should be Ly6G, and the related text in the legend should read “Numbers adjacent to outlined areas indicate Ly6G+CD11b+ cells (top row) or Ly6C+CD11b+ cells (bottom row).” The error has been corrected in the HTML and PDF versions of the article.
References
Medzhitov, R. Recognition of microorganisms and activation of the immune response. Nature 449, 819–826 (2007).
Kawai, T. & Akira, S. Innate immune recognition of viral infection. Nat. Immunol. 7, 131–137 (2006).
Burckstummer, T. et al. An orthogonal proteomic-genomic screen identifies AIM2 as a cytoplasmic DNA sensor for the inflammasome. Nat. Immunol. 10, 266–272 (2009).
Fernandes-Alnemri, T., Yu, J.W., Datta, P., Wu, J. & Alnemri, E.S. AIM2 activates the inflammasome and cell death in response to cytoplasmic DNA. Nature 458, 509–513 (2009).
Hornung, V. et al. AIM2 recognizes cytosolic dsDNA and forms a caspase-1-activating inflammasome with ASC. Nature 458, 514–518 (2009).
Roberts, T.L. et al. HIN-200 proteins regulate caspase activation in response to foreign cytoplasmic DNA. Science 323, 1057–1060 (2009).
Stetson, D.B. & Medzhitov, R. Type I interferons in host defense. Immunity 25, 373–381 (2006).
van den Broek, M.F., Muller, U., Huang, S., Zinkernagel, R.M. & Aguet, M. Immune defence in mice lacking type I and/or type II interferon receptors. Immunol. Rev. 148, 5–18 (1995).
Gilliet, M., Cao, W. & Liu, Y.J. Plasmacytoid dendritic cells: sensing nucleic acids in viral infection and autoimmune diseases. Nat. Rev. Immunol. 8, 594–606 (2008).
Kadowaki, N. et al. Subsets of human dendritic cell precursors express different toll-like receptors and respond to different microbial antigens. J. Exp. Med. 194, 863–869 (2001).
Yamamoto, M. et al. Role of adaptor TRIF in the MyD88-independent toll-like receptor signaling pathway. Science 301, 640–643 (2003).
Auerbuch, V., Brockstedt, D.G., Meyer-Morse, N., O'Riordan, M. & Portnoy, D.A. Mice lacking the type I interferon receptor are resistant to Listeria monocytogenes. J. Exp. Med. 200, 527–533 (2004).
O'Connell, R.M. et al. Type I interferon production enhances susceptibility to Listeria monocytogenes infection. J. Exp. Med. 200, 437–445 (2004).
Boehme, K.W., Guerrero, M. & Compton, T. Human cytomegalovirus envelope glycoproteins B and H are necessary for TLR2 activation in permissive cells. J. Immunol. 177, 7094–7102 (2006).
Compton, T. et al. Human cytomegalovirus activates inflammatory cytokine responses via CD14 and Toll-like receptor 2. J. Virol. 77, 4588–4596 (2003).
Szomolanyi-Tsuda, E., Liang, X., Welsh, R.M., Kurt-Jones, E.A. & Finberg, R.W. Role for TLR2 in NK cell-mediated control of murine cytomegalovirus in vivo. J. Virol. 80, 4286–4291 (2006).
Sato, A., Linehan, M.M. & Iwasaki, A. Dual recognition of herpes simplex viruses by TLR2 and TLR9 in dendritic cells. Proc. Natl. Acad. Sci. USA 103, 17343–17348 (2006).
Kurt-Jones, E.A. et al. Herpes simplex virus 1 interaction with Toll-like receptor 2 contributes to lethal encephalitis. Proc. Natl. Acad. Sci. USA 101, 1315–1320 (2004).
Chang, S., Dolganiuc, A. & Szabo, G. Toll-like receptors 1 and 6 are involved in TLR2-mediated macrophage activation by hepatitis C virus core and NS3 proteins. J. Leukoc. Biol. 82, 479–487 (2007).
Zhou, S. et al. MyD88 is critical for the development of innate and adaptive immunity during acute lymphocytic choriomeningitis virus infection. Eur. J. Immunol. 35, 822–830 (2005).
Bieback, K. et al. Hemagglutinin protein of wild-type measles virus activates toll-like receptor 2 signaling. J. Virol. 76, 8729–8736 (2002).
Zhu, J., Martinez, J., Huang, X. & Yang, Y. Innate immunity against vaccinia virus is mediated by TLR2 and requires TLR-independent production of IFN-beta. Blood 109, 619–625 (2007).
Toshchakov, V. et al. TLR4, but not TLR2, mediates IFN-β-induced STAT1α/β-dependent gene expression in macrophages. Nat. Immunol. 3, 392–398 (2002).
Kawai, T. et al. Lipopolysaccharide stimulates the MyD88-independent pathway and results in activation of IFN-regulatory factor 3 and the expression of a subset of lipopolysaccharide-inducible genes. J. Immunol. 167, 5887–5894 (2001).
Doyle, S. et al. IRF3 mediates a TLR3/TLR4-specific antiviral gene program. Immunity 17, 251–263 (2002).
Krug, A. et al. TLR9-dependent recognition of MCMV by IPC and DC generates coordinated cytokine responses that activate antiviral NK cell function. Immunity 21, 107–119 (2004).
Tabeta, K. et al. The Unc93b1 mutation 3d disrupts exogenous antigen presentation and signaling via Toll-like receptors 3, 7 and 9. Nat. Immunol. 7, 156–164 (2006).
Ablasser, A. et al. RIG-I-dependent sensing of poly(dA:dT) through the induction of an RNA polymerase III–transcribed RNA intermediate. Nat. Immunol. 10, 1065–1072 (2009).
Chiu, Y.-H., MacMillan, J.B. & Chen, Z.J. RNA polymerase III detects cytosolic DNA and induces type interferons through the RIG-I pathway. Cell 138, 576–591 (2009).
Asselin-Paturel, C. et al. Mouse type I IFN-producing cells are immature APCs with plasmacytoid morphology. Nat. Immunol. 2, 1144–1150 (2001).
Jin, M.S. et al. Crystal structure of the TLR1–TLR2 heterodimer induced by binding of a tri-acylated lipopeptide. Cell 130, 1071–1082 (2007).
Scheu, S., Dresing, P. & Locksley, R.M. Visualization of IFNβ production by plasmacytoid versus conventional dendritic cells under specific stimulation conditions in vivo. Proc. Natl. Acad. Sci. USA 105, 20416–20421 (2008).
Okusawa, T. et al. Relationship between structures and biological activities of mycoplasmal diacylated lipopeptides and their recognition by Toll-like receptors 2 and 6. Infect. Immun. 72, 1657–1665 (2004).
Cailhier, J.F. et al. Conditional macrophage ablation demonstrates that resident macrophages initiate acute peritoneal inflammation. J. Immunol. 174, 2336–2342 (2005).
Auffray, C., Sieweke, M.H. & Geissmann, F. Blood monocytes: development, heterogeneity, and relationship with dendritic cells. Annu. Rev. Immunol. 27, 669–692 (2009).
Geissmann, F., Jung, S. & Littman, D.R. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity 19, 71–82 (2003).
Serbina, N.V., Jia, T., Hohl, T.M. & Pamer, E.G. Monocyte-mediated defense against microbial pathogens. Annu. Rev. Immunol. 26, 421–452 (2008).
Kagan, J. et al. TRAM couples endocytosis of Toll-like receptor 4 to the induction of interferon-β. Nat. Immunol. 9, 361–368 (2008).
Lee, P.Y. et al. A novel type I IFN-producing cell subset in murine lupus. J. Immunol. 180, 5101–5108 (2008).
Krieg, A.M. Therapeutic potential of Toll-like receptor 9 activation. Nat. Rev. Drug Discov. 5, 471–484 (2006).
Ishii, K.J. et al. A Toll-like receptor-independent antiviral response induced by double-stranded B-form DNA. Nat. Immunol. 7, 40–48 (2006).
Stetson, D.B. & Medzhitov, R. Recognition of cytosolic DNA activates an IRF3-dependent innate immune response. Immunity 24, 93–103 (2006).
Takaoka, A. et al. DAI (DLM-1/ZBP1) is a cytosolic DNA sensor and an activator of innate immune response. Nature 448, 501–505 (2007).
Hayashi, F. et al. The innate immune response to bacterial flagellin is mediated by Toll-like receptor 5. Nature 410, 1099–1103 (2001).
Franchi, L. et al. Cytosolic flagellin requires Ipaf for activation of caspase-1 and interleukin 1-β in salmonella-infected macrophages. Nat. Immunol. 7, 576–582 (2006).
Miao, E.A. et al. Cytoplasmic flagellin activates caspase-1 and secretion of interleukin 1-β via Ipaf. Nat. Immunol. 7, 569–575 (2006).
Lightfield, K.L. et al. Critical function for Naip5 in inflammasome activation by a conserved carboxy-terminal domain of flagellin. Nat. Immunol. 9, 1171–1178 (2008).
Smith, K.D. et al. Toll-like receptor 5 recognizes a conserved site on flagellin required for protofilament formation and bacterial motility. Nat. Immunol. 4, 1247–1253 (2003).
Steven, A.C. & Spear, P.G. Biochemistry. Viral glycoproteins and an evolutionary conundrum. Science 313, 177–178 (2006).
Kurt-Jones, E.A. et al. Pattern recognition receptors TLR4 and CD14 mediate response to respiratory syncytial virus. Nat. Immunol. 1, 398–401 (2000).
Jude, B.A. et al. Subversion of the innate immune system by a retrovirus. Nat. Immunol. 4, 573–578 (2003).
Triantafilou, K. & Triantafilou, M. Coxsackievirus B4-induced cytokine production in pancreatic cells is mediated through toll-like receptor 4. J. Virol. 78, 11313–11320 (2004).
Georgel, P. et al. Vesicular stomatitis virus glycoprotein G activates a specific antiviral Toll-like receptor 4-dependent pathway. Virology 362, 304–313 (2007).
Jiang, Z. et al. CD14 is required for MyD88-independent LPS signaling. Nat. Immunol. 6, 565–570 (2005).
Crimmins, G.T. et al. Listeria monocytogenes multidrug resistance transporters activate a cytosolic surveillance pathway of innate immunity. Proc. Natl. Acad. Sci. USA 105, 10191–10196 (2008).
Ewald, S.E. et al. The ectodomain of Toll-like receptor 9 is cleaved to generate a functional receptor. Nature 456, 658–662 (2008).
Acknowledgements
We thank S. Akira (Osaka University) for Myd88−/−Trif−/− mice; D. Portnoy (University of California, Berkeley) for Irf3−/− and Ifnar1−/− mice and ISRE-L929 cells; K. Fitzgerald (University of Massachusetts) for Irf7−/− and Irf3−/−Irf7−/− mice; Z. Chen (University of Texas Southwestern Medical Center) for Mavs−/− mice; D. Raulet (University of California, Berkeley) for vaccinia virus (Western Reserve strain); L. Coscoy (University of California, Berkeley) for MCMV (Smith strain); M. Tokuyama, M. Fontana and K. Camfield for early contributions to this work; M. Mouchess for technical assistance; N. An (Infectious Disease BL2 Core of the Berkeley Center for Host-Pathogen Studies) for help with the production of vaccinia virus; D. Stetson (University of Washington) for key reagents; members of the Barton and Vance labs for discussions and advice; R. Vance and L. Coscoy for comments on the manuscript; and H. Nolla for assistance with flow cytometry. Supported by University of California, the Hellman Family Faculty Fund and the US National Institutes of Health (AI072429 to G.M.B., and GM007232 to R.B.).
Author information
Authors and Affiliations
Contributions
R.B. and G.M.B. designed the experiments; R.B. and L.L. did the experiments; R.M.L. provided reagents; and R.B. and G.M.B. wrote the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 4541 kb)
Rights and permissions
About this article
Cite this article
Barbalat, R., Lau, L., Locksley, R. et al. Toll-like receptor 2 on inflammatory monocytes induces type I interferon in response to viral but not bacterial ligands. Nat Immunol 10, 1200–1207 (2009). https://doi.org/10.1038/ni.1792
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1792
This article is cited by
-
Neutrophil extracellular traps contribute to immunothrombosis formation via the STING pathway in sepsis-associated lung injury
Cell Death Discovery (2023)
-
Investigation of TLR4 Antagonists for Prevention of Intestinal Inflammation
Inflammation (2023)
-
Creation of poxvirus expressing foot-and-mouth and peste des petits ruminant disease virus proteins
Applied Microbiology and Biotechnology (2023)
-
IFITM3 restricts virus-induced inflammatory cytokine production by limiting Nogo-B mediated TLR responses
Nature Communications (2022)
-
Pim1 promotes IFN-β production by interacting with IRF3
Experimental & Molecular Medicine (2022)