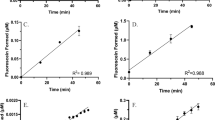
Abstract
Mechanism-based inactivators such as bergamottin are useful chemical tools for identifying the functions of specific active-site amino acid residues in the reactions catalyzed by cytochromes P450 (CYPs or P450s), which are responsible for the metabolism of a wide variety of drugs and endogenous substrates. In clinical settings, mechanism-based inactivation of P450s involved in xenobiotic metabolism has the potential to lead to adverse drug-drug interactions, and assays to identify and characterize drug candidates as P450 inactivators are important in drug discovery and development. Here we present a quantitative high-throughput protocol for investigating cytochrome P450 mechanism-based inactivators; we use the example of CYP2B6 and bergamottin to illustrate the finer points of this protocol. This protocol details the adaptation of a 7-ethoxytrifluoromethyl coumarin O-deethylation fluorescence activity assay to a 96-well microtiter plate format and uses a plate reader to detect the fluorescence of the product. Compared with previous methods, this protocol requires less P450 and takes significantly less time while greatly increasing throughput. The protocol as written takes ∼2 h to complete. The principles and procedures outlined in this protocol can be easily adapted to other inactivators, P450 isoforms, substrates and plate readers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Williams, A.J. et al. Drug-drug interactions for UDP-glucuronosyltransferase substrates: a pharmacokinetic explanation for typically observed low exposure (AUCI/AUC) ratios. Drug Metab. Dispos. 32, 1201–1208 (2004).
Porter, T.D. et al. Cytochrome P-450. Multiplicity of isoforms, substrates, and catalytic and regulatory mechanisms. J. Biol. Chem. 266, 13469–13472 (1991).
Evans, W.E. et al. Pharmacogenomics: translating functional genomics into rational therapeutics. Science 286, 487–491 (1999).
Kent, U.M. et al. Mechanism-based inactivators as probes of cytochrome P450 structure and function. Curr. Drug Metab. 2, 215–243 (2001).
Hollenberg, P.F. et al. Mechanism-based inactivation of human cytochrome P450s: experimental characterization, reactive intermediates, and clinical implications. Chem. Res. Toxicol. 21, 189–205 (2008).
Silverman, R.B. Mechanism-Based Enzyme Inactivation: Chemistry and Enzymology (CRC Press, Boca Raton, FL, 1988).
Mayer, R.T. et al. A real-time fluorescence assay for measuring N-dealkylation. Drug Metab. Dispos. 35, 103–109 (2007).
Blobaum, A.L. et al. P450 active site architecture and reversibility: inactivation of cytochromes P450 2B4 and 2B4 T302A by tert-butyl acetylenes. Biochemistry 44, 3831–3844 (2005).
Lin, H. et al. The grapefruit juice effect is not limited to cytochrome P450 3A4: evidence for bergamottin-dependent inactivation, heme destruction, and covalent binding to protein in P450s 2B6 and 3A5. J. Pharmacol. Exp. Ther. 313, 154–164 (2005).
Buters, J.T. et al. A highly sensitive tool for the assay of cytochrome P450 enzyme activity in rat, dog and man. Direct fluorescence monitoring of the deethylation of 7-ethoxy-4-trifluoromethylcoumarin. Biochem. Pharmacol. 46, 1577–1584 (1993).
DeLuca, J.G. et al. A direct, highly sensitive assay for cytochrome P-450 catalyzed O-deethylation using a novel coumarin analog. Biochem. Pharmacol. 37, 1731–1739 (1988).
Scott, E.E. et al. A truncation of 2B subfamily cytochromes P450 yields increased expression levels, increased solubility, and decreased aggregation while retaining function. Arch. Biochem. Biophys. 395, 57–68 (2001).
Rendic, S. Summary of information on human CYP enzymes: human P450 metabolism data. Drug Metab. Rev. 34, 83–448 (2002).
Xie, H.J. et al. Role of polymorphic human CYP2B6 in cyclophosphamide bioactivation. Pharmacogenomics J. 3, 53–61 (2003).
Schmiedlin-Ren, P. et al. Mechanisms of enhanced oral availability of CYP3A4 substrates by grapefruit constituents: decreased enterocyte CYP3A4 concentration and mechanism-based inactivation by furanocoumarins. Drug Metab. Dispos. 25, 1228–1233 (1997).
Bailey, D.G. et al. Grapefruit juice–drug interactions. Br. J. Clin. Pharmacol. 46, 101–110 (1998).
Bailey, D.G. et al. Grapefruit–felodipine interaction: effect of unprocessed fruit and probable active ingredients. Clin. Pharmacol. Ther. 68, 468–477 (2000).
Acknowledgements
This work was supported, in whole or in part, by National Institutes of Health Grant CA16954 (to P.F.H.).
Author information
Authors and Affiliations
Contributions
C.K. optimized and performed the assays with the advice of H.Z. C.K wrote the majority of the paper with the assistance of H.Z. and P.F.H.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Kenaan, C., Zhang, H. & Hollenberg, P. A quantitative high-throughput 96-well plate fluorescence assay for mechanism-based inactivators of cytochromes P450 exemplified using CYP2B6. Nat Protoc 5, 1652–1658 (2010). https://doi.org/10.1038/nprot.2010.125
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2010.125
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.