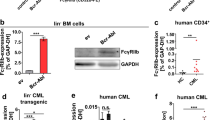
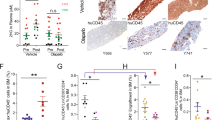
Abstract
Selective inhibition of the BCR/ABL tyrosine kinase by imatinib has become a first-line therapy for chronic myelogenous leukemia (CML). However, BCR/ABL-positive progenitors often persist despite treatment, and relapse associated with resistance to imatinib has been described in many patients with advanced disease. Drug efflux by P-glycoprotein (P-gp), as well as point mutations in BCR/ABL oncoprotein, has been implicated in the mechanism of resistance to imatinib. In this study, we established a murine transplantation model of CML-like myeloproliferative disease using Mdr1a/1b-null mice and analyzed the effects of loss of P-gp on resistance to imatinib. We found that mice transplanted with Mdr1a/1b-null bone marrow (BM) that had been transduced with a BCR/ABL retroviral vector displayed similar responses to imatinib, compared with those transplanted with BCR/ABL-transduced wild-type BM. In the absence of P-gp, the incidence and latency of disease in secondary recipients was not changed in imatinib-treated mice, relative to wild-type controls. Furthermore, K562 cells engineered to overexpress P-gp remained sensitive to imatinib-induced growth inhibition and cell death. Together, our findings suggest that P-gp expression in hematopoietic stem cells does not significantly contribute to imatinib resistance in CML.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Goldman JM, Melo JV . Chronic myeloid leukemia – advances in biology and new approaches to treatment. N Engl J Med 2003; 349: 1451–1464.
Druker BJ, Talpaz M, Resta DJ, Peng B, Buchdunger E, Ford JM et al. Efficacy and safety of a specific inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med 2001; 344: 1031–1037.
O'Brien SG, Guilhot F, Larson RA, Gathmann I, Baccarani M, Cervantes F, et al, IRIS Investigators. Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med 2003; 348: 994–1004.
Sawyers CL, Hochhaus A, Feldman E, Goldman JM, Miller CB, Ottmann OG et al. Imatinib induces hematologic and cytogenetic responses in patients with chronic myelogenous leukemia in myeloid blast crisis: results of a phase II study. Blood 2002; 99: 3530–3539.
Talpaz M, Silver RT, Druker BJ, Goldman JM, Gambacorti-Passerini C, Guilhot F et al. Imatinib induces durable hematologic and cytogenetic responses in patients with accelerated phase chronic myeloid leukemia: results of a phase II study. Blood 2002; 99: 1928–1937.
Bhatia R, Holtz M, Niu N, Gray R, Snyder DS, Sawyers CL et al. Persistence of malignant hematopoietic progenitors in chronic myelogenous leukemia patients in complete cytogenetic remission following imatinib mesylate treatment. Blood 2003; 101: 4701–4707.
Mahon FX, Deininger MW, Schultheis B, Chabrol J, Reiffers J, Goldman JM et al. Selection and characterization of BCR-ABL positive cell lines with differential sensitivity to the tyrosine kinase inhibitor STI571: diverse mechanisms of resistance. Blood 2000; 96: 1070–1079.
Gorre ME, Mohammed M, Ellwood K, Hsu N, Paquette R, Rao PN et al. Clinical resistance to STI-571 cancer therapy caused by BCR-ABL gene mutation or amplification. Science 2001; 293: 876–880.
Gambacorti-Passerini C, Zucchetti M, Russo D, Frapolli R, Verga M, Bungaro S et al. Alpha 1 acid glycoprotein binds to imatinib (STI571) and substantially alters its pharmacokinetics in chronic myeloid leukemia patients. Clin Cancer Res 2003; 9: 625–632.
Shah NP, Nicoll JM, Nagar B, Gorre ME, Paquette RL, Kuriyan J et al. Multiple BCR-ABL kinase domain mutations confer polyclonal resistance to the tyrosine kinase inhibitor imatinib (STI571) in chronic phase and blast crisis chronic myeloid leukemia. Cancer Cell 2002; 2: 117–125.
Azam M, Latek RR, Daley GQ . Mechanisms of autoinhibition and STI571/imatinib resistance revealed by mutagenesis of BCR-ABL. Cell 2003; 112: 831–843.
Mahon FX, Belloc F, Lagarde V, Chollet C, Moreau-Gaudry F, Reiffers J et al. MDR1 gene overexpression confers resistance to imatinib mesylate in leukemia cell line models. Blood 2003; 101: 2368–2373.
Che XF, Nakajima Y, Sumizawa T, Ikeda R, Ren XQ, Zheng CL et al. Reversal of P-glycoprotein mediated multidrug resistance by a newly synthesized 1,4-benzothiazipine derivative, JTV-519. Cancer Lett 2002; 187: 111–119.
Kotaki M, Motoji T, Takanashi M, Wang YH, Mizoguchi H . Anti-proliferative effect of the abl tyrosine kinase inhibitor STI571 on the P-glycoprotein positive K562/ADM cell line. Cancer Lett 2003; 199: 61–68.
Illmer T, Schaich M, Platzbecker U, Freiberg-Richter J, Oelschlagel U, von Bonin M et al. P-glycoprotein-mediated drug efflux is a resistance mechanism of chronic myelogenous leukemia cells to treatment with imatinib mesylate. Leukemia 2004; 18: 401–408.
Hegedus T, Orfi L, Seprodi A, Varadi A, Sarkadi B, Keri G . Interaction of tyrosine kinase inhibitors with the human multidrug transporter proteins, MDR1 and MRP1. Biochim Biophys Acta 2002; 1587: 318–325.
Mukai M, Che XF, Furukawa T, Sumizawa T, Aoki S, Ren XQ et al. Reversal of the resistance to STI571 in human chronic myelogenous leukemia K562 cells. Cancer Sci 2003; 94: 557–563.
Dai H, Marbach P, Lemaire M, Hayes M, Elmquist WF . Distribution of STI-571 to the brain is limited by P-glycoprotein-mediated efflux. J Pharmacol Exp Ther 2003; 304: 1085–1092.
Chaudhary PM, Roninson IB . Expression and activity of P-glycoprotein, a multidrug efflux pump, in human hematopoietic stem cells. Cell 1991; 66: 85–94.
Sorrentino BP, McDonagh KT, Woods D, Orlic D . Expression of retroviral vectors containing the human multidrug resistance 1 cDNA in hematopoietic cells of transplanted mice. Blood 1995; 86: 491–501.
Ferrao PT, Frost MJ, Siah SP, Ashman LK . Overexpression of P-glycoprotein in K562 cells does not confer resistance to the growth inhibitory effects of imatinib (STI571) in vitro. Blood 2003; 102: 4499–4503.
Schinkel AH, Mayer U, Wagenaar E, Mol CA, van Deemter L, Smit JJ et al. Normal viability and altered pharmacokinetics in mice lacking mdr1-type (drug-transporting) P-glycoproteins. Proc Natl Acad Sci USA 1997; 94: 4028–4033.
Bunting KD, Galipeau J, Topham D, Benaim E, Sorrentino BP . Transduction of murine bone marrow cells with an MDR1 vector enables ex vivo stem cell expansion, but these expanded grafts cause a myeloproliferative syndrome in transplanted mice. Blood 1998; 92: 2269–2279.
Mayotte N, Roy DC, Yao J, Kroon E, Sauvageau G . Oncogenic interaction between BCR-ABL and NUP98-HOXA9 demonstrated by the use of an in vitro purging culture system. Blood 2002; 100: 4177–4184.
Persons DA, Mehaffey MG, Kaleko M, Nienhuis AW, Vanin EF . An improved method for generating retroviral producer clones for vectors lacking a selectable marker gene. Blood Cells Mol Dis 1998; 24: 167–182.
Bunting KD, Zhou S, Lu T, Sorrentino BP . Enforced P-glycoprotein pump function in murine bone marrow cells results in expansion of side population stem cells in vitro and repopulating cells in vivo. Blood 2000; 96: 902–909.
Wolff NC, Ilaria Jr RL . Establishment of a murine model for therapy-treated chronic myelogenous leukemia using the tyrosine kinase inhibitor STI571. Blood 2001; 98: 2808–2816.
Pear WS, Miller JP, Xu L, Pui JC, Soffer B, Quackenbush RC et al. Efficient and rapid induction of a chronic myelogenous leukemia-like myeloproliferative disease in mice receiving P210 bcr/abl-transduced bone marrow. Blood 1998; 92: 3780–3792.
Zhang X, Ren R . Bcr-Abl efficiently induces a myeloproliferative disease and production of excess interleukin-3 and granulocyte-macrophage colony-stimulating factor in mice: a novel model for chronic myelogenous leukemia. Blood 1998; 92: 3829–3840.
Li S, Ilaria Jr RL, Million RP, Daley GQ, Van Etten RA . The P190, P210, and P230 forms of the BCR/ABL oncogene induce a similar chronic myeloid leukemia-like syndrome in mice but have different lymphoid leukemogenic activity. J Exp Med 1999; 189: 1399–1412.
Pardal R, Clarke MF, Morrison SJ . Applying the principles of stem-cell biology to cancer. Nat Rev Cancer 2003; 3: 895–902.
Zhou S, Schuetz JD, Bunting KD, Colapietro AM, Sampath J, Morris JJ et al. The ABC transporter Bcrp1/ABCG2 is expressed in a wide variety of stem cells and is a molecular determinant of the side-population phenotype. Nat Med 2001; 7: 1028–1034.
Zhou S, Morris JJ, Barnes Y, Lan L, Schuetz JD, Sorrentino BP . Bcrp1 gene expression is required for normal numbers of side population stem cells in mice, and confers relative protection to mitoxantrone in hematopoietic cells in vivo. Proc Natl Acad Sci USA 2002; 99: 12339–12344.
Burger H, van Tol H, Boersma AW, Brok M, Wiemer EA, Stoter G et al. Imatinib mesylate (STI571) is a substrate for the breast cancer resistance protein (BCRP)/ABCG2 drug pump. Blood 2004; 104: 2940–2942.
Breedveld P, Pluim D, Cipriani G, Wielinga P, van Tellingen O, Schinkel AH et al. The effect of Bcrp1 (Abcg2) on the in vivo pharmacokinetics and brain penetration of imatinib mesylate (Gleevec): implications for the use of breast cancer resistance protein and P-glycoprotein inhibitors to enable the brain penetration of imatinib in patients. Cancer Res 2005; 65: 2577–2582.
Graham SM, Jorgensen HG, Allan E, Pearson C, Alcorn MJ, Richmond L et al. Primitive, quiescent, Philadelphia-positive stem cells from patients with chronic myeloid leukemia are insensitive to STI571 in vitro. Blood 2002; 99: 319–325.
Ren R . Mechanisms of BCR-ABL in the pathogenesis of chronic myelogenous leukaemia. Nat Rev Cancer 2005; 5: 172–183.
Houghton PJ, Germain GS, Harwood FC, Schuetz JD, Stewart CF, Buchdunger E et al. Imatinib mesylate is a potent inhibitor of the ABCG2 (BCRP) transporter and reverses resistance to topotecan and SN-38 in vitro. Cancer Res 2004; 64: 2333–2337.
Zhou S, Zong Y, Lu T, Sorrentino BP . Hematopoietic cells from mice that are deficient in both Bcrp1/Abcg2 and Mdr1a/1b develop normally but are sensitized to mitoxantrone. Biotechniques 2003; 35: 1248–1252.
Acknowledgements
We thank Flow Cytometry Lab and Animal Diagnostic Lab for sample analysis. This study is supported by NIH Grant R01 HL67366 (to BPS), by the American Lebanese Syrian Associated Charities and by the Hal and Alma Reagan Fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zong, Y., Zhou, S. & Sorrentino, B. Loss of P-glycoprotein expression in hematopoietic stem cells does not improve responses to imatinib in a murine model of chronic myelogenous leukemia. Leukemia 19, 1590–1596 (2005). https://doi.org/10.1038/sj.leu.2403853
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403853
Keywords
This article is cited by
-
Chronic Myeloid Leukemia CD34+ cells have reduced uptake of imatinib due to low OCT-1 Activity
Leukemia (2010)
-
Extending the duration of response in chronic myelogenous leukemia: targeted therapy with sequential tyrosine kinase inhibitors
Oncology Reviews (2009)
-
Resistance reversal by RNAi silencing of MDR1 in CML cells associated with increase in imatinib intracellular levels
Leukemia (2007)
-
Reply to ‘Resistance reversal by RNAi silencing of MDR1 in CML cells associated with increase in imatinib intracellular levels’ by Widmer et al.
Leukemia (2007)
-
ATP-binding-cassette transporters in hematopoietic stem cells and their utility as therapeutical targets in acute and chronic myeloid leukemia
Leukemia (2007)