Abstract
Linagliptin is an orally active small-molecule inhibitor of dipeptidyl peptidase (DPP)-4, which was first licensed in the US, Europe, Japan and other territories in 2011 to improve glycaemic control in adults with type 2 diabetes mellitus. Linagliptin is the first and thus far the only DPP-4 inhibitor, and oral anti-hyperglycaemic drug in general, to be approved as a single-strength once-daily dose (5 mg). Compared with other available DPP-4 inhibitors, linagliptin has a unique pharmacokinetic/pharmacodynamic profile that is characterized by target-mediated nonlinear pharmacokinetics, concentration-dependent protein binding, minimal renal clearance and no requirements for dose adjustment for any intrinsic or extrinsic factor.
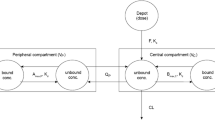
After single or multiple oral doses of 1–10 mg, linagliptin displays less than dose-proportional increases in maximum plasma concentration (Cmax) and area under the plasma concentration-time curve (AUC). Linagliptin is rapidly absorbed after oral administration, with Cmax occurring after approximately 90 minutes, and reaches steady-state concentrations within 4 days. With the therapeutic dose, steady-state Cmax (11–12nmol/L) and AUC (∼150nmol · h/L) are approximately 1.3-fold greater than after a single dose, indicating little drug accumulation with repeat dosing. Linagliptin exhibits concentration-dependent protein binding in human plasma in vitro (99% at 1 nmol/L to 75–89% at >30 nmol/L) and has a large apparent volume of distribution, demonstrating extensive distribution into tissues. The nonlinear pharmacokinetics of linagliptin are best described by a two-compartmental model that incorporates target-mediated drug disposition resulting from high-affinity, saturable binding to DPP-4. The oral bioavailability of linagliptin estimated with this model is approximately 30%. Linagliptin has a long terminal half-life (>100 hours); however, its accumulation half-life is much shorter (approximately 10 hours), accounting for the rapid attainment of steady state. The majority of linagliptin is eliminated as parent compound, demonstrating that metabolism plays a minor role in the overall pharmacokinetics in humans. The main, pharmacologically inactive S-3-hydroxypiperidinyl metabolite accounted for approximately 17% of the total drug-related compounds in plasma. Linagliptin is eliminated primarily in faeces, with only around 5% of the oral therapeutic dose excreted in the urine at steady state. Linagliptin potently inhibits DPP-4 (inhibition constant 1 nmol/L), and trough drug concentrations achieved with therapeutic dosing inhibit >80% of plasma DPP-4 activity, the threshold associated with maximal antihyperglycaemic effects in animal models.
There are no clinically relevant alterations in linagliptin pharmacokinetics resulting from renal impair-ment, hepatic impairment, coadministration with food, race, body weight, sex or age. In vitro, linagliptin is a weak substrate and weak inhibitor of cytochrome P450 (CYP) 3 A4 and permeability glycoprotein (P-gp) but not of other CYP isozymes or ATP-binding cassette transporters. Clinical studies have revealed no relevant drug interactions when coadministered with other drugs commonly prescribed to patients with type 2 diabetes, including the narrow therapeutic index drugs warfarin and digoxin. Linagliptin plasma exposure is reduced by potent inducers of CYP3A4 or P-gp. Linagliptin has demonstrated a large safety window (>100-fold the recommended daily dose) and clinically relevant antihyperglycaemic effects in patients with type 2 diabetes.








Similar content being viewed by others
References
Danaei G, Finucane MM, Lu Y, et al. National, regional, and global trends in fasting plasma glucose and diabetes prevalence since 1980: systematic analysis of health examination surveys and epidemiological studies with 370 country-years and 2.7 million participants. Lancet 2011 Jul 2; 378(9785): 31–40
Shaw JE, Sicree RA, Zimmet PZ. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract 2010; 87(1): 4–14
Centers for Disease Control and Prevention. National diabetes fact sheet: national estimates and general information on diabetes and prediabetes in the United States, 2011 [online]. Available from URL: http://www.cdc.gov/diabetes/pubs/pdf/ndfs_2007.pdf [Accessed 2010 Sep 10]
Turner RC, Cull CA, Frighi V, et al. Glycemic control with diet, sulfonylurea, metformin, or insulin in patients with type 2 diabetes mellitus: progressive requirement for multiple therapies (UKPDS 49). UK Prospective Diabetes Study (UKPDS) Group. JAMA 1999 Jun 2; 281(21): 2005–12
Defronzo RA. Banting lecture: from the triumvirate to the ominous octet: a new paradigm for the treatment of type 2 diabetes mellitus. Diabetes 2009 Apr; 58(4): 773–95
Nathan DM, Buse JB, Davidson MB, et al. Medical management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 2009 Jan; 32(1): 193–203
Handelsman Y, Mechanick JI, Blonde L, et al. American Association of Clinical Endocrinologists Medical Guidelines for Clinical Practice for developing a diabetes mellitus comprehensive care plan. Endocr Pract 2011 Mar–Apr; 17 Suppl. 2: 1–53
Kreymann B, Williams G, Ghatei MA, et al. Glucagon-like peptide-1 7-36: a physiological incretin in man. Lancet 1987 Dec 5; 2(8571): 1300–4
Dupre J, Ross SA, Watson D, et al. Stimulation of insulin secretion by gastric inhibitory polypeptide in man. J Clin Endocrinol Metab 1973 Nov; 37(5): 826–8
Baggio LL, Drucker DJ. Biology of incretins: GLP-1 and GIP. Gastroenterology 2007 May; 132(6): 2131–57
Mentlein R, Gallwitz B, Schmidt WE. Dipeptidyl-peptidase IV hydrolyses gastric inhibitory polypeptide, glucagon-like peptide-1 (7–36)amide, peptide histidine methionine and is responsible for their degradation in human serum. Eur J Biochem 1993 Jun 15; 214(3): 829–35
Tradjenta (linagliptin) prescribing information. Ridgefield (CT): Boehringer Ingelheim Pharmaceuticals Inc., 2010
Data on file, Boehringer Ingelheim, 2005
Blech S, Ludwig-Schwellinger E, Grafe-Mody EU, et al. The metabolism and disposition of the oral dipeptidyl peptidase-4 inhibitor, linagliptin, in humans. Drug Metab Dispos 2010 Apr; 38(4): 667–78
Data on file, Boehringer Ingelheim, 2006
Thomas L, Eckhardt M, Langkopf E, et al. (R)-8-(3-amino-piperidin- 1-yl)-7-but-2-ynyl-3-methyl-1-(4-methyl-quinazolin-2-ylmethyl)-3,7-dihydro-purine-2,6-dione (BI1356), a novel xanthine-based dipeptidyl peptidase 4 inhibitor, has a superior potency and longer duration of action compared with other dipeptidyl peptidase-4 inhibitors. J Pharmacol Exp Ther 2008 Apr; 325(1): 175–82
Lankas GR, Leiting B, Roy RS, et al. Dipeptidyl peptidase IV inhibition for the treatment of type 2 diabetes: potential importance of selectivity over dipeptidyl peptidases 8 and 9. Diabetes 2005 Oct; 54(10): 2988–94
Burkey BF, Hoffmann PK, Hassiepen U, et al. Adverse effects of dipeptidyl peptidases 8 and 9 inhibition in rodents revisited. Diabetes Obes Metab 2008 Nov; 10(11): 1057–61
Schurmann C, Linke A, Engelmann-Pilger K, et al. The dipeptiayl peptidase-4 inhibitor linagliptin attenuates inflammation and accelerates epithelialization in wounds of diabetic ob/ob mice. J Pharmacol Exp Ther. Epub 2012 Apr 4
Lambeir AM, Durinx C, Scharpe S, et al. Dipeptidyl-peptidase IV from bench to bedside: an update on structural properties, functions, and clinical aspects of the enzyme DPP IV. Crit Rev Clin Lab Sci 2003 Jun; 40(3): 209–94
Fuchs H, Tillement JP, Urien S, et al. Concentration-dependent plasma protein binding of the novel dipeptidyl peptidase 4 inhibitor BI1356 due to saturable binding to its target in plasma of mice, rats and humans. J Pharm Pharmacol 2009 Jan; 61(1): 55–62
Retlich S, Withopf B, Greischel A, et al. Binding to dipeptidyl peptidase-4 determines the disposition of linagliptin (BI 1356): investigations in DPP-4 deficient and wildtype rats. Biopharm Drug Dispos 2009 Nov; 30(8): 422–36
Fuchs H, Binder R, Greischel A. Tissue distribution of the novel DPP-4 inhibitor BI 1356 is dominated by saturable binding to its target in rats. Biopharm Drug Dispos 2009 Jul; 30(5): 229–40
Hüttner S, Graefe-Mody EU, Withopf B, et al. Safety, tolerability, pharmacokinetics, and pharmacodynamics of single oral doses of BI 1356, an inhibitor of dipeptidyl peptidase 4, in healthy male volunteers. J Clin Pharmacol 2008 Oct; 48(10): 1171–8
Retlich S, Duval V, Ring A, et al. Pharmacokinetics and pharmacodynamics of single rising intravenous doses (0.5 mg–10 mg) and determination of absolute bioavailability of the dipeptidyl peptidase-4 inhibitor linagliptin (BI 1356) in healthy male subjects. Clin Pharmacokinet 2010 Dec 1; 49(12): 829–40
Graefe-Mody U, Giessmann T, Ring A, et al. A randomized, open-label, crossover study evaluating the effect of food on the relative bioavailability of linagliptin in healthy subjects. Clin Ther 2011 Aug; 33(8): 1096–103
Sarashina A, Sesoko S, Nakashima M, et al. Linagliptin, a dipeptidyl peptidase-4 inhibitor in development for the treatment of type 2 diabetes mellitus: a phase I, randomized, double-blind, placebo-controlled trial of single and multiple escalating doses in healthy adult male Japanese subjects. Clin Ther 2010 Jun; 32(6): 1188–204
Friedrich C, Glund S, Lionetti D, et al. Pharmacokinetic and pharmacodynamic evaluation of linagliptin in African American patients with type 2 diabetes [abstract]. J Clin Pharmacol 2011 Sep; 51(9): 1336
Heise T, Graefe-Mody EU, Huttner S, et al. Pharmacokinetics, pharmacodynamics and tolerability of multiple oral doses of linagliptin, a dipeptidyl peptidase-4 inhibitor in male type 2 diabetes patients. Diabetes Obes Metab 2009 Aug; 11(8): 786–94
Forst T, Uhlig-Laske B, Ring A, et al. The oral DPP-4 inhibitor linagliptin significantly lowers HbA1c after 4 weeks of treatment in patients with type 2 diabetes mellitus. Diabetes Obes Metab 2011 Jun; 13(6): 542–50
Horie Y, Kanada S, Watada H, et al. Pharmacokinetic, pharmacodynamic, and tolerability profiles of the dipeptidyl peptidase-4 inhibitor linagliptin: a 4-week multicenter, randomized, double-blind, placebo-controlled phase IIa study in Japanese type 2 diabetes patients. Clin Ther 2011 Jul; 33(7): 973–89
Retlich S, Duval V, Graefe-Mody U, et al. Impact of target-mediated drug disposition on Linagliptin pharmacokinetics and DPP-4 inhibition in type 2 diabetic patients. J Clin Pharmacol 2010 Aug; 50(8): 873–85
Januvia (sitagliptin) prescribing information. Whitehouse Station (NJ): Merck and Co. Inc., 2010
Onglyza (saxagliptin) prescribing information. Princeton (NJ): Bristol-Myers Squibb Company, 2010
Galvus (vildagliptin) prescribing information. Camberley, Surrey, UK: No-vartis Pharmaceuticals UK Ltd, 2010
Christopher R, Covington P, Davenport M, et al. Pharmacokinetics, pharmacodynamics, and tolerability of single increasing doses of the dipeptidyl peptidase-4 inhibitor alogliptin in healthy male subjects. Clin Ther 2008 Mar; 30(3): 513–27
Data on file, Boehringer Ingelheim, 2010
Trajenta (linagliptin). Summary of product characteristics. Ingelheim: Boehringer Ingelheim International GmbH, 2011
Trazenta (linagliptin) prescribing information. Tokyo: Nippon Boehringer Ingelheim Co., Ltd, 2011
Finding a safe and effective dose of linagliptin in paediatric patients with type 2 diabetes [ClinicalTrials.gov identifier NCT01342484]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://www.clinicaltrials.gov [Accessed 2011 Dec 16]
Efficacy and safety of linagliptin in elderly patients with type 2 diabetes [ClinicalTrials.gov identifier NCT01084005]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://www.clinicaltrials.gov [Accessed 2011 Dec 16]
Kim D, Wang L, Beconi M, et al. (2R)-4-oxo-4-[3-(trifluoromethyl)-5,6-dihy-dro[1,2,4]triazolo[4,3-a]pyrazin-7(8H)-yl]-1-(2,4,5-trifluorophenyl)butan-2-amine: a potent, orally active dipeptidyl peptidase IV inhibitor for the treatment of type 2 diabetes. J Med Chem 2005 Jan 13; 48(1): 141–51
Data on file, Boehringer Ingelheim, 2009
Del Prato S, Barnett AH, Huisman H, et al. Effect of linagliptin monotherapy on glycaemic control and markers of β-cell function in patients with inadequately controlled type 2 diabetes: a randomized controlled trial. Diabetes Obes Metab 2011 Mar; 13(3): 258–67
Taskinen MR, Rosenstock J, Tamminen I, et al. Safety and efficacy of linagliptin as add-on therapy to metformin in patients with type 2 diabetes: a randomized, double-blind, placebo-controlled study. Diabetes Obes Metab 2010 Jan; 13(1): 65–74
Owens DR, Swallow R, Dugi KA, et al. Efficacy and safety of linagliptin in persons with type 2 diabetes inadequately controlled by a combination of metformin and sulphonylurea: a 24-week randomized study. Diabet Med 2011 Nov; 28(11): 1352–61
Kawamori R, Inagaki N, Araki E, et al. Linagliptin monotherapy provides superior glycaemic control versus placebo or voglibose with comparable safety in Japanese patients with type 2 diabetes: a randomized, placebo and active comparator-controlled, double-blind study. Diabetes Obes Metab 2012 Apr; 14(4): 348–57
Monami M, Iacomelli I, Marchionni N, et al. Dipeptidyl peptidase-4 inhibitors in type 2 diabetes: a meta-analysis of randomized clinical trials. Nutr Metab Cardiovasc Dis 2010 May; 20(4): 224–35
Bergman AJ, Stevens C, Zhou Y, et al. Pharmacokinetic and pharmacodynamic properties of multiple oral doses of sitagliptin, a dipeptidyl peptidase-IV inhibitor: a double-blind, randomized, placebo-controlled study in healthy male volunteers. Clin Ther 2006 Jan; 28(1): 55–72
Richter B, Bandeira-Echtler E, Bergerhoff K, et al. Dipeptidyl peptidase-4 (DPP-4) inhibitors for type 2 diabetes mellitus. Cochrane Database Syst Rev 2008; (2): CD006739
Arechavaleta R, Seck T, Chen Y, et al. Efficacy and safety of treatment with sitagliptin or glimepiride in patients with type 2 diabetes inadequately controlled on metformin monotherapy: a randomized, double-blind, non-inferiority trial. Diabetes Obes Metab 2011 Feb; 13(2): 160–8
Matthews DR, Dejager S, Ahren B, et al. Vildagliptin add-on to metformin produces similar efficacy and reduced hypoglycaemic risk compared with glimepiride, with no weight gain: results from a 2-year study. Diabetes Obes Metab 2010 Sep; 12(9): 780–9
Gallwitz B, Rosenstock R, Rauch T, et al. Two-year efficacy and safety of linagliptin compared with glimepiride in patients with type 2 diabetes inadequately controlled on metformin: a randomised, double-blind, active controlled, non-inferiority trial. Lancet. In Press
Cernea S, Raz I. Therapy in the early stage: incretins. Diabetes Care 2011 May; 34 Suppl. 2: S264–71
Ring A, Port A, Graefe-Mody EU, et al. The DPP-4 inhibitor linagliptin does not prolong the QT interval at therapeutic and supratherapeutic doses. Br J Clin Pharmacol 2011 Jul; 72(1): 39–50
Graefe-Mody U, Friedrich C, Port A, et al. Effect of renal impairment on the pharmacokinetics of the dipeptidyl peptidase-4 inhibitor linagliptin. Diabetes Obes Metab 2011 Oct; 13(10): 939–46
Graefe-Mody EU, Rose P, Ring A, et al. Pharmacokinetics of linagliptin in subjects with hepatic impairment. Br J Clin Pharmacol. Epub 2012 Jan 13
Friedrich C, Emser A, Woerle H-J, et al. Renal impairment has no relevant effect on long-term exposure of linagliptin in patients with type 2 diabetes mellitus [poster]. Diabetes 2011 Jul; 60 Suppl. 1: A303
Koro CE, Lee BH, Bowlin SJ. Antidiabetic medication use and prevalence of chronic kidney disease among patients with type 2 diabetes mellitus in the United States. Clin Ther 2009 Nov; 31(11): 2608–17
Meyers JL, Candrilli SD, Kovacs B. Type 2 diabetes mellitus and renal impairment in a large outpatient electronic medical records database: rates of diagnosis and antihyperglycemic medication dose adjustment. Postgrad Med 2011 May; 123(3): 133–43
Sloan L, Newman J, Sauce C, et al. Safety and efficacy of linagliptin in type 2 diabetes patients with severe renal impairment [poster]. Diabetes 2011 Jul; 60 Suppl. 1: A114
Newman J, McGill J, Patel S, et al. Long-term efficacy and safety of linagliptin in patients with type 2 diabetes and severe renal impairment [poster]. Diabetologia 2011; 54 Suppl. 1: S108
Graefe-Mody EU, Padula S, Ring A, et al. Evaluation of the potential for steady-state pharmacokinetic and pharmacodynamic interactions between the DPP-4 inhibitor linagliptin and metformin in healthy subjects. Curr Med Res Opin 2009 Aug; 25(8): 1963–72
Graefe-Mody U, Rose P, Ring A, et al. Assessment of the pharmacokinetic interaction between the novel DPP-4 inhibitor linagliptin and a sulfonylurea, glyburide, in healthy subjects. Drug Metab Pharmacokinet 2011; 26(2): 123–9
Graefe-Mody EU, Jungnik A, Ring A, et al. Evaluation of the pharmacokinetic interaction between the dipeptidyl peptidase-4 inhibitor linagliptin and pioglitazone in healthy volunteers. Int J Clin Pharmacol Ther 2010 Oct; 48(10): 652–61
Graefe-Mody U, Huettner S, Stahle H, et al. Effect of linagliptin (BI 1356) on the steady-state pharmacokinetics of simvastatin. Int J Clin Pharmacol Ther 2010 Jun; 48(6): 367–74
Friedrich C, Ring A, Brand T, et al. Evaluation of the pharmacokinetic interaction after multiple oral doses of linagliptin and digoxin in healthy volunteers. Eur J Drug Metab Pharmacokinet 2011 Mar; 36(1): 17–24
Food and Drug Administration. Guidance for Industry. Drug interaction studies: study design, data analysis, and implications for dosing and labeling. Rockville (MD): US Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER). September 2006 [online]. Available from URL: http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM072101.pdf [Accessed 2011 Oct 7]
Graefe-Mody EU, Brand T, Ring A, et al. Effect of linagliptin on the pharmacokinetics and pharmacodynamics of warfarin in healthy volunteers. Int J Clin Pharmacol Ther 2011 May; 49(5): 300–10
Friedrich C, Port A, Ring A, et al. Effect of multiple oral doses of linagliptin on the steady-state pharmacokinetics of a combination oral contraceptive in healthy female adults: an open-label, two-period, fixed-sequence, multiple-dose study. Clin Drug Investig 2011; 31(9): 643–53
de Heer J, Holst JJ. Sulfonylurea compounds uncouple the glucose dependence of the insulinotropic effect of glucagon-like peptide 1. Diabetes 2007 Feb; 56(2): 438–43
Johansen OE, Neubacher D, von Eynatten M, et al. Cardiovascular safety with linagliptin in patients with type 2 diabetes mellitus: a pre-specified, prospective, and adjudicated meta-analysis of a phase 3 programme. Cardiovasc Diabetol 2012 Jan 10; 11: 3
CAROLINA: Cardiovascular Outcome Study of Linagliptin Versus Glime-piride in Patients With Type 2 Diabetes [ClinicalTrials.gov identifier NCT01243424]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://www.clinicaltrials.gov [Accessed 2011 Dec 16]
Acknowledgements
This work was supported by Boehringer Ingelheim Pharma GmbH & Co. KG, the manufacturer of linagliptin. All authors are employees of Boehringer Ingelheim. Medical writing assistance was provided by Giles Brooke, PhD, of Envision Scientific Solutions during the preparation of this paper and was supported by Boehringer Ingelheim. The authors were fully responsible for all content and editorial decisions, and were involved at all stages of manuscript development.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Graefe-Mody, U., Retlich, S. & Friedrich, C. Clinical Pharmacokinetics and Pharmacodynamics of Linagliptin. Clin Pharmacokinet 51, 411–427 (2012). https://doi.org/10.2165/11630900-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11630900-000000000-00000