Summary
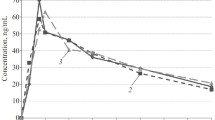
Comparative pharmacokinetic studies with the β-receptor blocking drugs propranolol, metoprolol, sotalol and atenolol, differing greatly in lipophilicity, and their main route of elimination were performed in light-dark-synchronized rats after equimolar single (6 μmoles/kg) or multiple (6x6 μmoles/kg) drug application. Drug concentrations were determined in plasma and various target organs of the drugs, e.g. heart, muscle, lung and brain, after drug application in the light period (L) and dark period (D), respectively. After single drug administration pharmacokinetic parameters of all drugs depended on the L and D conditions. Elimination half-lives in plasma and organs were shorter during D than during L. No L-D-differences were found in initial drug concentrations of the hydrophilic drugs sotalol and atenolol. In contrast, C0-values of the lipophilic propranolol in highly perfused organs (muscle, lung, brain) and of metoprolol in muscle tissue were significantly higher in D than in L. No obvious temporal dependency was found in other pharmacokinetic parameters (AUC, plasma clearance,V dβ) with the exception inV dβ of propranolol. Due to the different physico-chemical properties of the compounds inter-drug-differences in pharmacokinetic parameters including drug accumulation into lung and brain tissue were observed. Multiple drug dosing abolished the circadian-phase-dependency in the elimination half-lives of the drugs due to an increase in D. Only for the highly lipophilic propranolol half-lives in highly perfused organs were still shorter in D than in L. It is concluded that L-D-differences in drug half-lives after single dose application are mainly due to circadian variations in drug elimination with a higher hepatic (propranolol, metoprolol) or renal (sotalol, atenolol) elimination in the activity period of rats during D. Additional studies with propranolol on heart rate of conscious rats revealed that a maximum in β-receptor blockade was achieved at 10 μmoles/kg in L but not in D. Thus, it is assumed that abolition of circadian-phase-dependency in half-lives after 6x6 μmoles/kg of the drugs may be due to the longer lasting and more pronounced β-receptor blockade after multiple drug dosing over a period of several hours in D. Thereby, liver-flow-dependent elimination of propranolol and metoprolol and renal elimination of sotalol and atenolol is reduced to base-line levels found in L.
Similar content being viewed by others
References
Åblad B, Borg KO, Carlsson E, Ek L, Johnsson G, Malmfors T, Regardh C-G (1973) A survey of the pharmacological properties of metoprolol in animals and man. Acta Pharmacol Toxicol 36 Suppl V:7–23
Appelgren C, Borg KO, Elofsson R, Johansson KA (1974) Binding of adrenergic beta-receptor antagonists to human serum albumin. Acta Pharmacol Suecica 11:325–332
Borg KO, Fellenius E, Johansson R, Wallborg M (1975) Pharmacokinetic studies of metoprolol-(3H) in the rat and the dog. Acta Pharmacol Toxicol 36 Suppl V:104–115
Branch RA, Shand DG, Wilkinson GR, Nies AS (1973) The reduction of lidocaine clearance by d,l-propranolol: An example of hemodynamic drug interaction. J Pharmacol Exp Ther 184: 515–519
Brown HC, Carruthers SG, Johnston GD, Kelly JG, McAinsh J, McDevitt DG, Shanks RG (1976) Clinical pharmacologic observations on atenolol, a beta-adrenoceptor blocker. Clin Pharmacol Ther 20:524–534
Cambar J, Lemoigne F, Toussaint Ch (1979) Diurnal variations evidence for glomerular filtration in the rat. Experientia 35:1607–1609
Cohn C, Webb L, Joseph D (1970) Diurnal rhythm in urinary electrolyte excretions by the rat: Influence of feeding habits. Life Sci 9:803–809
Edvinsson L, Nielsen KC, Owman Ch (1973) Circadian rhythm in cerebral blood volume of mouse. Experientia 29:432–433
Evans GH, Wilkinson GR, Shand DG (1973) The disposition of propranolol. IV. A dominant role for tissue uptake in the dose dependent extraction of propranolol by the perfused rat liver. J Pharmacol Exp Ther 186:447–454
Farmer JB, Levy GP (1968) A simple method for recording the electrocardiogram and heart rate from conscious animals. Brit J Pharmacol Chemother 32:193–200
Halberg E, Halberg F, Carandente F (1981) From autopsy or biopsy to the physiologist's chronopsy. Chronobiologia 8:145–159
Hellenbrecht D, Lemmer B, Wiethold G, Grobecker H (1973) Measurement of hydrophobicity, surface activity, local anasthesia, and myocardial conduction velocity as quantitative parameters of the non-specific membrane affinity of nine β-adrenergic blocking agents. Naunyn-Schmiedeberg's Arch Pharmacol 277: 211–226
Hilfenhaus M (1976) Circadian rhythm of the renin-angiotensin-aldosteron system in the rat. Arch Toxicol 36:305–316
Jori A, DiSalle E, Santini V (1971) Daily rhythmic variation and liver drug metabolism in rats. Biochem Pharmacol 20:2964–2969
Lemmer B (1981) Pharmacokinetics of β-adrenoceptor blocking drugs of different polarity (propranolol, metoprolol, atenolol) in plasma and various organs of the light-dark-synchronized rat. Naunyn-Schmiedeberg's Arch Pharmacol 316:R60
Lemmer B, Bathe K (1982) Stereospecific and circadian-phase-dependent kinetic behavior ofd,l-,l-, andd-propranolol in plasma, heart and brain of light-dark-synchronized rats. J Cardiovasc Pharmacol 4:635–644
Lemmer B, Charrier A (1980) Antagonism by chlorisondamine and propranolol, but not by atenolol, of the circadian-phase-dependent phentolamine-induced changes in the cardiac noradrenaline turnover in the rat. Naunyn-Schmiedeberg's Arch Pharmacol 313:205–212
Lemmer B, Lang P-H (1984a) Evidence for a circadian-phase-dependency in3H-dihydroalprenolol binding and cyclic AMP content in heart ventricles of L-D-synchronized rats. Pol J Pharmacol Pharm 36:275–280
Lemmer B, Lang P-H (1984b) Circadian-phase-dependency in3H-diphydroalprenolol binding to rat heart ventricular membranes. Chronobiol Int 1:217–223
Lemmer B, Saller R (1974) Influence of light and darkness on the turnover of noradrenaline in the rat heart. Naunyn-Schmiedeberg's Arch Pharmacol 282:75–84
Lemmer B, Weimer R (1983) Differences in the content and turnover of noradrenaline in rat heart atria and ventricles and circadian-phase-dependency. Experientia 39:998–1000
Lemmer B, Simrock R, Hellenbrecht D, Smolensky MH (1980) Chronopharmacological studies with propranolol in rodents: Implications for the management of COPD patients with cardiovascular disease. In: Smolensky MH, McGovern JP, Reinberg A (eds) Recent advances in the chronobiology of allergy and immunology. Pergamon Press, Oxford New York, pp 195–208
Lemmer B, Winkler H, Fink M (1983a) Comparative pharmacokinetics of propranolol, metoprolol and atenolol after single and multiple dosing in the rat. Naunyn-Schmiedeberg's Arch Pharmacol 322:R33
Lemmer B, Bathe K, Lang PH, Neumann G, Winkler H (1983b) Chronopharmacology of β-adrenoceptor blocking drugs: Pharmacokinetic and pharmacodynamic studies in rats. J Am Coll Toxicol 2:347–358
Lemmer B, Ohm T, Winkler H (1984) Determination of the beta-adrenoceptor blocking drug sotalol in plasma and tissues of the rat by high-performance liquid chromatography with ultra-violet detection. J Chromatogr 309:187–192
Malini PL; Stocchi E, Ambrosioni E (1983) Propranolol and renal hemodynamic at rest and during exercis. Int J Clin Pharmacol Ther Toxicol 21:611–614
Menzel W (1940) Über einen 24-Stunden-Rhythmus im Blutkreislauf des Menschen. Acta Med Scand Suppl 108:166–177
Nair V, Casper R (1969) The influence of light on daily rhythm in hepatic drug metabolizing enzyms. Life Sci 8:1291–1298
Nies AS, Evans GH, Shand DG (1973a) The hemodynamic effects of beta-adrenergic blockade on the flow-dependent hepatic clearance of propranolol. J Pharmacol Exp Ther 184:716–720
Nies AS, Evans GH, Shand DG (1973b) Regional hemodynamic effects of beta-adrenergic blockade with propranolol in the unanesthetized primate. Amer Heart J 85:97–102
North C, Feuers RJ, Scheving LE, Pauly JE, Tsai TH, Casciana DA (1981) Circadian organization of thirteen liver and six brain enzymes of the mouse. Amer J Anat 162:183–199
Pirttiaho HI, Sotaniemi FA, Pelkonen RO, Pitkänen U, Anttila M, Sundquist H (1980) Roles of hepatic blood flow and enzyme activity in the kinetics of propranolol and sotalol. Brit J Clin Pharmacol 9:399–405
Radzialowski FM, Bousquet WF (1967) Circadian rhythm in hepatic drug metabolizing activity in the rat. Life Sci 6:2545–2548
Schnelle K, Garrett ER (1973) Pharmacokinetics of the β-adrenergic blocker sotalol in dogs. J Pharmacol Sci 62:362–375
Shand DG, Rangno RE, Evans GH (1972) The disposition of propranolol. II. Hepatic elimination in the rat. Pharmacology 8:344–352
Smith DF (1973) Diurnal variations of lithium clearance in the rat. Int Pharmacopsychiat 8:9–103
Sotaniemi EA, Anttila M, Pelkonen RD, Jarvensivu P, Sundquist H (1979) Plasma clearance of propranolol and sotalol and hepatic drug-metabolizing enzyme activity. Clin Pharmacol Ther 26:151–161
Winkler H, Ried W, Lemmer B (1982) High-performance liquid chromatographic method for the quantitative analysis of the aryloxypropanolamines propranolol, metoprolol and atenolol in plasma and tissue. J Chromatogr 226:223–234
Woods PB, Robinson ML (1981) An investigation of the comparative liposolubilities of β-adrenoceptor blocking agents. J Pharm Pharmacol 33:172–173
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lemmer, B., Winkler, H., Ohm, T. et al. Chronopharmacokinetics of beta-receptor blocking drugs of different lipophilicity (propranolol, metoprolol, sotalol, atenolol) in plasma and tissues after single and multiple dosing in the rat. Naunyn-Schmiedeberg's Arch. Pharmacol. 330, 42–49 (1985). https://doi.org/10.1007/BF00586708
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00586708