Summary
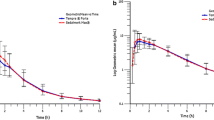
The bioavailability and pharmacokinetics of theophylline from a plain uncoated and 2 newly designed, sustained-release tablet formulations, as compared to intravenous aminophylline, were studied in 12 healthy adult male volunteers. The subjects were divided into two groups (n=6) with respect to smoking habit and on 4 separate occasions each received, on a randomized cross-over basis, a single dose of 400 mg equivalent of theophylline from every dosage form. The intravenous aminophylline study showed that habitual smoking had a significant (p<0.05) effect on plasma theophylline clearance (0.051±0.006 vs 0.035±0.004 l/kg/h). Smoking significantly reduced the raw AUC from the 4 dosage forms (p<0.05), but did not change the characteristics of absorption of each formulation. There was a non-significant trend towards reduced absolute bioavailability of theophylline from sustained-release formulations in smokers (percentage mean difference — 16% for one formulation and 13% for another). The trend was not observed for the plain uncoated tablet, which was rapidly absorbed (p<0.01 to 0.05 in Ka, tmax and Cmax compared to sustained-release tablets). Similarity of the in vitro dissolution profiles of the two sustained-release formulations did not imply similarity of the in vivo absorption characteristics. Plasma clearances of theophylline and antipyrine were significantly correlated (p<0.05,r=0.693,n=10). Thus, smoking enhanced the elimination of theophylline regardless of the dosage form administered. However, the extent to which habitual smoking may affect the hepatic first-pass effect on theophylline from sustained-release formulations requires further study. The results also suggest that theophylline and antipyrine may share a similar or common and presumably polycyclic hydrocarbon-inducible form(s) of microsomal drugmetabolizing enzyme.
Similar content being viewed by others
References
Weinberger M, Hendeles L, Bighley L (1978) The relation of product formulation to absorption of oral theophylline. N Engl J Med 299: 852–857
Spangler DL, Kalof DD, Bloom FL, Witting HJ (1978) Theophylline bioavailability following oral administration of six sustained-release preparations. Ann Allergy 40: 6–11
Bell T, Bigley J (1978) Sustained-release theophylline therapy for chronic childhood asthma. Pediatrics 62: 352–358
Kelly HW, Murphy S (1980) Efficacy of a 12-hour sustained-release preparation in maintaining therapeutic serum theophylline levels in asthmatic children. Pediatrics 66: 97–102
Weinberger M, Hendeles L, Wong L (1981) Relationship of formulation of dosing interval of fluctuation of serum theophylline concentration in children with asthma. J Pediatr 99: 145–152
Dasta J, Mirtallo JM, Altman M (1979) Comparison of standard- and sustained-release theophylline tablets in patients with chronic obstructive pulmonary disease. Am J Hosp Pharm 36: 613–617
Hunt SN, Jusko WJ, Yurchak AM (1976) Effect of smoking on theophylline disposition. Clin Pharmacol Ther 19: 546–551
Jenne J, Nagasawa H, McHugh R, MacDonald F, Wyse E (1975) Decreased theophylline half-life in cigarette smokers. Life Sci 17: 195–198
Powell JR, Thiercelin J-F, Vozeh S, Sansom L, Riegelman S (1977) The influence of cigarette smoking and sex on theophylline disposition. Am Rev Respir Dis 116: 17–23
Grygiel JJ, Birkett DJ (1981) Cigarette smoking and theophylline clearance and metabolism. Clin Pharmacol Ther 30: 491–496
Jusko WJ (1978) Role of tobacco smoking in pharmacokinetics. J Pharmacokinet Biopharm 6: 7–39
Ogilvie RI (1978) Clinical pharmacokinetics of theophylline. Clin Pharmacokinet 3: 267–293
Jusko WJ, Gardner MJ, Mangione A, Schentag JJ, Koup JR, Vance JW (1979) Factors affecting theophylline clearances: Age, tobacco, marijuana, cirrhosis, congestive heart failure, obesity, oral contraceptives, benzodiazepines, barbiturates and ethanol. J Pharm Sci 68: 1358–1366
Adams RF, Bandemark FL, Schmidt GJ (1976) More sensitive high-pressure liquid-chromatographic determination of theophylline in serum. Clin Chem 22: 1903–1906
Ishizaki T, Watanabe M, Morishita N (1979) The effect of assay methods on plasma levels and pharmacokinetics of theophylline: HPLC and EIA. Br J Clin Pharmacol 7: 333–341
Metzler CM, Elfring GL, McEven AJ (1974) A package of computer programs for pharmacokinetic modeling. Biometrics 30: 562–563
Wagner JG (1976) Linear pharmacokinetic equations allowing direct calculations of many needed pharmacokinetic parameters from the coefficients and exponents of poly-exponential equations which have been fitted to the data. J Pharmacokinet Biopharm 4: 443–467
Boxenbaum HG, Riegelman S, Elashoff RM (1974) Statistical estimations in pharmacokinetics. J Pharmacokinet Biopharm 2: 123–148
Loo JCK, Riegelman S (1970) Assessment of pharmacokinetic constants from postinfusion blood curves obtained after i.v. infusion. J Pharm Sci 59: 53–55
Gibaldi M, Perrier D (1975) Pharmacokinetics. 1st ed. Marcel Dekker, New York, p 37
Prescott LF, Adjepon-Yamoah KK, Roberts E (1973) Rapid gas-liquid chrohatographic estimation of antipyrine in plasma. J Pharm Pharmacol 25: 205–207
Fixley M, Shen DD, Azarnoff DL (1977) Theophylline bioavailability. A comparison of the oral absorption of a theophylline elixir and two combination theophylline tablets to intravenous aminophylline. Am Rev Respir Dis 115: 955–962
Upton RA, Sansom L, Guentert TW, Powell JR, Thiercelin J-F, Shah VP, Coates PE, Riegelman S (1980) Evaluation of the absorption from 15 commercial theophylline products indicating deficiencies in currently applied bioavailability criteria. J Pharmacokinet Biopharm 8: 229–242
Hendeles L, Weinberger M, Bighley L (1977) Absolute bioavailability of oral theophylline. Am J Hosp Pharm 34: 525–527
Jonkman JHG, Berg WC, de Vries K, de Zeeuw RA, Schoenmaker R, Grimberg N (1981) Disposition and clinical pharmacokinetics of theophylline after administration of a new sustained release tablet. Eur J Clin Pharmacol 21: 39–44
Mitenko PA, Ogilvie RI (1974) Bioavailability and efficacy of a sustained-release theophylline tablet. Clin Pharmacol Ther 16: 720–726
Kappas A, Alvares AP, Anderson KE, Pantuck EJ, Pantuck CB, Chang R, Conney AH (1978) Effect of charcoal-broiled beef on antipyrine and theophylline metabolism. Clin Pharmacol Ther 23: 445–450
Vestal RE, Norris AH, Tobin JD, Cohen BH, Shock NW, Andres R (1975) Antipyrine metabolism in man: Influence of age, alcohol, caffeine, and smoking. Clin Pharmacol Ther 18: 425–432
Hart P, Farrell GC, Cooksley WGE, Powell LW (1976) Enhanced drug metabolism in cigarette smokers. Br Med J 2: 147–149
Landay RA, Gonzalez MA, Taylor JC (1978) Effect of phenobarbital on theophylline disposition. J Allergy Clin Immunol 62: 27–29
Kampffmeyer HG (1971) Elimination of phenacetin and phenazone by man before and after treatment with phenobarbital. Eur J Clin Pharmacol 3: 113–118
Roberts CJC, Jackson L, Halliwell M, Branch RA (1976) The relationship between liver volume, antipyrine clearance and indocyanine green clearance before and after phenobarbitone administration in man. Br J Clin Pharmacol 3: 907–913
Roberts RK, Grice J, Wood L, Petroff V, McGuffie C (1981) Cimetidine impairs the elimination of theophylline and antipyrine. Gastroenterology 81: 19–21
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Horai, Y., Ishizaki, T., Sasaki, T. et al. Bioavailability and pharmacokinetics of theophylline in plain uncoated and sustained-release dosage forms in relation to smoking habit I. Single dose study. Eur J Clin Pharmacol 24, 79–87 (1983). https://doi.org/10.1007/BF00613931
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00613931