Summary
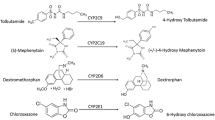
Pharmacokinetic gender-dependent differences in cytochrome P450-mediated drug metabolism, especially CYP3A4, and their clinical implications are increasingly apparent. CYP3A4 seems to be the most important CYP isoform in both bioactivation and N-dechloroethylation of the alkylating prodrug ifosfamide, but informations about possible gender-related differences are lacking. Therefore we compared in 10 male and 10 female liver microsomal preparations the contents and activities of specific isoenzymes, involved in both metabolic pathways, especially CYP3A4, further CYP2A6, CYP2C9 and CYP2B6 and measured the in vitro activities of these microsomes in the ifosfamide 4-hydroxylation and N-dechloroethylation using high-sensitive HPLC/MS and--UV detection methods.
Statistically significant differences between male and female livers were found in the mean CYP3A4 contents and activities. These differences had no consequences on the ifosfamide 4-hydroxylation activities of liver microsomes in vitro. In contrast, in the ifosfamide N-dechloroethylation reaction we found a statistically significant difference between the liver microsomes of male and female patients (0.13 ± 0.05 nmol/min · nmolP450 vs. 0.28 ± 0.13 nmol/min · nmolP450, respectively).
In conclusion, we firstly demonstrated such gender-related difference in the ifosfamide N-dechloroethylation, which could result in a higher risk of partly severe neurotoxic side effects in female patients.
Similar content being viewed by others
References
Tanaka E (1999) Gender-related differences in pharmacokinetics and their significance. J Clin Pharm Ther 24: 339–346.
Gleiter CH and Gundert-Remy U (1996) Gender differences in pharmacokinetics. Eur J Drug Metab Pharmacokinet 21: 123–128.
Kahan BD, Kramer WG, Wideman C, Flechner SM, Lorber MI and Van Buren CT (1986) Demographic factors affecting the pharmacokinetics of cyclosporine estimated by radioimmunoassay. Transplantation 41: 459–464.
Holazo AA, Winkler MB and Patel IH (1988) Effects of age, gender and oral contraceptives on intramuscular midazolam pharmacokinetics. J Clin Pharmacol 28: 1040–1045.
Austin KL, Mather LE, Philpot CR and McDonald PJ (1980) Intersubject and dose-related variability after intravenous administration of erythromycin. Br J Clin Pharmacol 10: 273–279.
Watkins PB, Turgeon DK, Saenger P, Lown KS, Kolars JC, Hamilton T, Fishman K, Guzelian PS and Voorhees JJ (1992) Comparison of urinary 6-beta-cortisol and the erythromycin breath test as measures of hepatic P450IIIA (CYP3A) activity. Clin Pharmacol Ther 52: 265–273.
Hunt CM, Westerkan WR and Stave GM (1992) Effect of age and gender on the activity of human hepatic CYP3A. Biochem Pharmacol 44: 275–283.
Beierle I, Meibohm B and Derendorf H (1999) Gender differences in pharmacokinetics and pharmacodynamics. Int J Clin Pharmacol Ther 37: 529–547.
Weber GF and Waxman DJ (1993) Activation of the anti-cancer drug ifosphamide by rat liver microsomal P450 enzymes. Biochem Pharmacol 45: 1685–1694.
Wainer IW, Ducharme J and Granvil CP (1996) The N-dechloroethylation of ifosfamide: using stereochemistry to obtain an accurate picture of a clinically relevant metabolic pathway. Cancer Chemother Pharmacol 37: 332–336.
Brüggemann SK, Kisro J and Wagner T (1997) Ifosfamide cytotoxicity on human tumor and renal cells: Role of chloroacetaldehyde in comparison to 4-hydroxyifosfamide. Cancer Res 57: 2676–2680.
Börner K, Kisro J, Brüggemann SK, Hagenah W, Peters SO and Wagner T (2000) Metabolism of ifosfamide to chloroacetaldehyde contributes to antitumor activity in vivo. Drug Metab Dispos 28: 573–576.
Roy P, Yu LY, Crespi CL and Waxman DJ (1999) Development of a substrate-activity based approach to identify the major human liver P-450 catalysts of cyclophosphamide and ifosfamide activation based on cDNA-expressed activities and liver microsomal P-450 profiles. Drug Metab Dispos 27: 655–666.
Walker D, Flinois JP, Monkman SC, Beloc C, Boddy AV, Cholerton S, Daly AK, Lind MJ, Pearson AD, Beaune PH and Idle JR (1994) Identification of the major human hepatic ccytochrome P450 involved in activation and N-dechloroethylation of ifosfamide. Biochem Pharmacol 47: 1157–1163.
Roy P, Tretyakov O, Wright J and Waxman DJ (1999) Stereoselective metabolism of ifosfamide by human P450s 3A4 and 2B6. Favorable metabolic properties ofR-enantiomer. Drug Metab Dispos 27: 1309–1318.
Granvil CP, Madan A, Sharkawi M, Parkinsonn A and Wainer I (1999) Role of CYP2B6 and CYP3A4 in the vitro N-dechloroethylation of (R)-and (S)-ifosfamide in human liver microsomes. Drug Metab Dispos 27: 533–541.
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the prinziple of proteindye binding. Anal Biochem 72: 248–254.
Omura T and Sato R (1964) The carbon monoxide-binding pigment of liver microsomes. J Biol Chem 239: 2370–2378.
Miles JS, McLaren AW, Forrester LM, Glancey MJ, Lang MA and Wolf CR (1990) Identificationn of the human liver cytochrome P-450 responsible for coumarin 7-hydroxylase activity. Biochemm J 267: 365–371.
Relling MV, Aoyama T, Gonzalez FJ and Meyer UA (1990) Tolbutamide and mephenytoin hydroxylation by human cytochrome P450s in the CYP2C subfamily. J Pharmacol Exp Ther 252: 442–447.
Sanwald P, Blankson EA, Dulery BD, Schoun J, Huebert ND and Dow J (1995) Isocratic high-performance liquid chromatographic method for the separation of testosterone metabolites. J Chromatogr B — Biomed Appl 672: 207–215.
Ruzicka JA and Ruenitz PC (1992) Cytochrome P450-mediated N-dechloroethylation of cyclophosphamide and ifosfamide in the rat. Drug Metab Dispos 20: 770–772.
Masurel D, Houghton PJ, Young CL and Wainer IW (1990) Efficacy, toxicity, pharmacokinetics, and in vitro metabolism of the enantiomers of ifosfamide inn mice. Cancer Res 50: 252–255.
Harris RZ, Benet LZ and Scchwartz JB (1995) Gender effects in pharmacokinetics and pharmacodynamics. Drugs 50: 222–239.
Pollock BG (1997) Gender differences in psychotropic drug metabolism. Psychopharmacol Bull 33: 235–241.
Huang Z, Roy P and Waxman DJ (2000) Role of human liver microsomal CYP3A4 and CYP2B6 in catalyzing N-dechloroethylation of cyclophosphamide and ifosfamide. Biochem Pharmacol 59: 961–972.
Wagner T and Drings P (1987) Pharmacokinetics and bioavailability of oral ifosfamide. Contr Oncol 26: 53–59.
Manegold C, Bischoff H, Fischer JR, Löchner S, Peukert M, Schmähl A and Drings P (1992) Oral ifosfamide-mesna: a clinical investigation in advanced non-small cell lung cancer. Ann Oncol 3: 723–726.
Anderson NR and Tandon DS (1991) Ifosfamide extrapyramidal neurotoxicity. Cancer 68: 72–75.
DiMaggio JR, Brown R, Baile WF and Schapira D (1994) Hallucinations and ifosfamide-induced neurotoxicity. Cancer 73: 1509–1514.
Merimsky O, Reider-Groswasser I, Wigler N and Chaitchik S (1992) Encephalopathy in ifosfamide-treated patients. Acta Neurol Scand 86: 5821–525
Watkin SW, Husband DJ, Green JA and Warenius HM (1989) Ifosfamide encephalopathy: a reappraisal. Eur J Cancer Clin Oncol 25: 1303–1310.
Küpfer A, Aeschlimann C and Cerny T (1996) Methylene blue and the neurotoxic mechanisms of ifosfamide enccephalopathy. Eur J Clin Pharmacol 50: 249–252.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schmidt, R., Baumann, F., Hanschmann, H. et al. Gender difference in ifosfamide metabolism by human liver microsomes. Eur. J. Drug Metab. Pharmacokinet. 26, 193–200 (2001). https://doi.org/10.1007/BF03190396
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03190396