Abstract
Purpose
To investigate the pharmacokinetics, metabolism and tolerability of afatinib (BIBW 2992), an oral irreversible ErbB family blocker, in healthy male volunteers.
Methods
In this open-label, single-center study, 8 healthy male volunteers received a single oral dose of 15 mg [14C]-radiolabeled afatinib (equivalent to 22.2 mg of the dimaleinate salt) as a solution. Blood, urine and fecal samples were collected for at least 96 hours (h) after dosing. Plasma and urine concentrations of afatinib were analyzed using high-performance liquid chromatography–tandem mass spectrometry. [14C]-radioactivity levels in plasma, whole blood, urine and feces were measured by liquid scintillation counting methods. Metabolite patterns were assessed by high-performance liquid chromatography.
Results
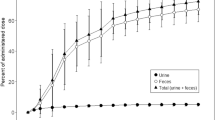
[14C]-radioactivity was mainly excreted via feces (85.4%). Overall recovery of [14C]-radioactivity was 89.5%, indicative of a complete mass balance. Afatinib was slowly absorbed, with maximum plasma concentrations achieved at a median of 6 h after dosing, declining thereafter in a biexponential manner. The geometric mean terminal half-life of afatinib was 33.9 h in plasma and longer for [14C]-radioactivity in plasma and whole blood. Apparent total body clearance for afatinib was high (geometric mean 1,530 mL/min). The high volume of distribution (4,500 L) in plasma may indicate a high tissue distribution. Afatinib was metabolized to only a minor extent, with the main metabolite afatinib covalently bound to plasma proteins. Oxidative metabolism mediated via cytochrome P-450 was of negligible importance for the elimination of afatinib. Afatinib was well tolerated.
Conclusions
Afatinib displayed a complete mass balance with the main route of excretion via feces. Afatinib undergoes minimal metabolism.






Similar content being viewed by others
References
Li D, Ambrogio L, Shimamura T, Kubo S, Takahashi M, Chirieac LR, Padera RF, Shapiro GI, Baum A, Himmelsbach F, Rettig WJ, Meyerson M, Solca F, Greulich H, Wong KK (2008) BIBW2992, an irreversible EGFR/HER2 inhibitor highly effective in preclinical lung cancer models. Oncogene 27(34):4702–4711
Bahleda R, Soria J, Berge Y, Massard C, Wind S, Uttenreuther-Fischer MM, Fleischer F, De-Montserrat H, Solca F, Tschoepe I, Delord J (2011) Phase I trial assessing safety and pharmacokinetics of afatinib (BIBW 2992) with intravenous weekly vinorelbine in advanced solid tumors (abstract). J Clin Oncol 29(15 Suppl):2585
Agus DB, Terlizzi E, Stopfer P, Amelsberg A, Gordon MS (2006) A phase I dose escalation study of BIBW 2992, an irreversible dual EGFR/HER2 receptor tyrosine kinase inhibitor, in a continuous schedule in patients with advanced solid tumours (abstract 2074). J Clin Oncol 24(18 Suppl):97S
Lewis N, Marshall J, Amelsberg A, Cohen RB, Stopfer P, Hwang J, Malik S (2006) A phase I dose escalation study of BIBW 2992, an irreversible dual EGFR/HER2 receptor tyrosine kinase inhibitor, in a 3 week on 1 week off schedule in patients with advanced solid tumors (abstract 3091). J Clin Oncol 24(18 Suppl):143S
Eskens FA, Mom CH, Planting AS, Gietema JA, Amelsberg A, Huisman H, van Doorn L, Burger H, Stopfer P, Verweij J, de Vries EG (2008) A phase I dose escalation study of BIBW 2992, an irreversible dual inhibitor of epidermal growth factor receptor 1 (EGFR) and 2 (HER2) tyrosine kinase in a 2-week on, 2-week off schedule in patients with advanced solid tumours. Br J Cancer 98(1):80–85
Yap TA, Vidal L, Adam J, Stephens P, Spicer J, Shaw H, Ang J, Temple G, Bell S, Shahidi M, Uttenreuther-Fischer M, Stopfer P, Futreal A, Calvert H, de Bono JS, Plummer R (2010) Phase I trial of the irreversible EGFR and HER2 kinase inhibitor BIBW 2992 in patients with advanced solid tumors. J Clin Oncol 28(25):3965–3972
Ang J, Mikropoulos C, Stavridi F, Rudman S, Uttenreuther-Fisher M, Shahidi M, Pemberton K, Wind S, de Bono J, Spicer JF (2009) A phase I study of daily BIBW 2992, an irreversible EGFR/HER-2 dual kinase inhibitor, in combination with weekly paclitaxel (abstract). J Clin Oncol 27(15):e14541
Awada AH, Dumez H, Wolter P, Hendlisz A, Besse-Hammer T, Piccart M, Uttenreuther-Fischer M, Stopfer P, Taton M, Schoffski P (2009) A phase I dose finding study of the 3-day administration of BIBW 2992, an irreversible dual EGFR/HER-2 inhibitor, in combination with three-weekly docetaxel in patients with advanced solid tumors (abstract). J Clin Oncol 27(15 Suppl):3556
Vermorken JB, Machiels JH, Rottey S, Thurm HC, Pelling K, Lahogue A (2010) Phase Ib study evaluating the combination of BIBW 2992 with two different standard chemotherapy regimens, cisplatin/paclitaxel (PT) and cisplatin/5-FU (PF), in patients with advanced solid tumors (abstract). J Clin Oncol 28(15 Suppl):e13521
Miller VA, Hirsh V, Cadranel J, Chen Y-M, Park K, Kim S-W, Caicun Z, Oberdick M, Shahidi M, Yang C-H (2010) Phase IIB/III double-blind randomized trial of afatinib (BIBW 2992, an irreversible inhibitor of EGFR/HER1 and HER2) + best supportive care (BSC) versus placebo 1 + BSC in patients with NSCLC failing 1-2 lines of chemotherapy and erlotinib or gefitinib (LUX-LUNG 1) [abstract LBA1]. Ann Oncol 21(Suppl 8):viii1
Yang CH, Shih JY, Su WC, Hsia TC, Sequist LV, Chang GC, Calvo R, Cong XJ, Shahidi M, Miller VA (2010) A Phase II study of afatinib (BIBW 2992) in patients with adenocarcinoma of the lung and activating EGFR/HER1 mutations (LUX-Lung 2) [abstract O-042]. J Thorac Oncol 5(12):S376–S377
Hickish T, Wheatley D, Lin N, Carey L, Houston S, Mendelson D, Solca F, Uttenreuther-Fischer M, Jones H, Winer E (2009) Use of BIBW 2992, a novel irreversible EGFR/HER1, HER2 tyrosine kinase inhibitor to treat patients with HER2-Positive metastatic breast cancer after failure of treatment with trastuzumab (abstract 5060). Cancer Res 69(24):785S
Ranson M, Hammond LA, Ferry D, Kris M, Tullo A, Murray PI, Miller V, Averbuch S, Ochs J, Morris C, Feyereislova A, Swaisland H, Rowinsky EK (2002) ZD1839, a selective oral epidermal growth factor receptor-tyrosine kinase inhibitor, is well tolerated and active in patients with solid, malignant tumors: results of a phase I trial. J Clin Oncol 20(9):2240–2250
Baselga J, Rischin D, Ranson M, Calvert H, Raymond E, Kieback DG, Kaye SB, Gianni L, Harris A, Bjork T, Averbuch SD, Feyereislova A, Swaisland H, Rojo F, Albanell J (2002) Phase I safety, pharmacokinetic, and pharmacodynamic trial of ZD1839, a selective oral epidermal growth factor receptor tyrosine kinase inhibitor, in patients with five selected solid tumor types. J Clin Oncol 20(21):4292–4302
Hidalgo M, Bloedow D (2003) Pharmacokinetics and pharmacodynamics: maximizing the clinical potential of Erlotinib (Tarceva). Semin Oncol 30(3 Suppl 7):25–33
Burris HA 3rd, Hurwitz HI, Dees EC, Dowlati A, Blackwell KL, O’Neil B, Marcom PK, Ellis MJ, Overmoyer B, Jones SF, Harris JL, Smith DA, Koch KM, Stead A, Mangum S, Spector NL (2005) Phase I safety, pharmacokinetics, and clinical activity study of lapatinib (GW572016), a reversible dual inhibitor of epidermal growth factor receptor tyrosine kinases, in heavily pretreated patients with metastatic carcinomas. J Clin Oncol 23(23):5305–5313
McKillop D, Hutchison M, Partridge EA, Bushby N, Cooper CM, Clarkson-Jones JA, Herron W, Swaisland HC (2004) Metabolic disposition of gefitinib, an epidermal growth factor receptor tyrosine kinase inhibitor, in rat, dog and man. Xenobiotica 34(10):917–934
Ling J, Johnson KA, Miao Z, Rakhit A, Pantze MP, Hamilton M, Lum BL, Prakash C (2006) Metabolism and excretion of erlotinib, a small molecule inhibitor of epidermal growth factor receptor tyrosine kinase, in healthy male volunteers. Drug Metab Dispos 34(3):420–426
Wang J, Li-Chan XX, Atherton J, Deng L, Espina R, Yu L, Horwatt P, Ross S, Lockhead S, Ahmad S, Chandrasekaran A, Oganesian A, Scatina J, Mutlib A, Talaat R (2010) Characterization of HKI-272 covalent binding to human serum albumin. Drug Metab Dispos 38(7):1083–1093
Song X, Varker H, Eichelbaum M, Stopfer P, Shahidi M, Wilson K, Kaiser R, Finnern HW (2011) Treatment of lung cancer patients and concomitant use of drugs interacting with cytochrome P450 isoenzymes. Lung Cancer 74(1):103–111
Acknowledgments
This study was supported by Boehringer Ingelheim. Editorial assistance was provided with funding from Boehringer Ingelheim.
Conflict of interest
All authors are employees of Boehringer Ingelheim.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stopfer, P., Marzin, K., Narjes, H. et al. Afatinib pharmacokinetics and metabolism after oral administration to healthy male volunteers. Cancer Chemother Pharmacol 69, 1051–1061 (2012). https://doi.org/10.1007/s00280-011-1803-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-011-1803-9