Abstract
Abstract
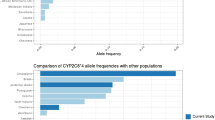
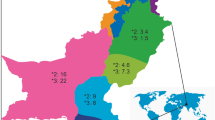
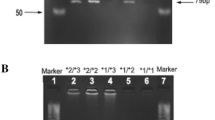
This study aimed to investigate the allele frequencies of CYP2B6 gene in 193 Han Chinese and compared with 91 Uygur Chinese. Five single nucleotide polymorphisms of CYP2B6, 64C>T, 516G>T, 777C>A, 785A>G and 1459C>T, were tested using the polymerase chain reaction–restriction fragment length polymorphism method. The allele frequencies for CYP2B6*2, CYP2B6*3, CYP2B6*4, CYP2B6*5, CYP2B6*6, CYP2B6*7 and CYP2B6*9 in Han and Uygur Chinese were 0.034 and 0.027, 0 and 0.011, 0.091 and 0.033, 0.003 and 0.049, 0.184 and 0.214, 0 and 0.022, and 0.018 and 0.044, respectively, with CYP2B6*4, CYP2B6*5, and CYP2B6*7 being significantly different between these two races (P<0.05). CYP2B6*6 was the most prevalent allele among all detected variants in Han and Uygur Chinese. The most frequent genotypes were CYP2B6*1/CYP2B6*1 (50.8%), CYP2B6*1/CYP2B6*6 (24.4%), and CYP2B6*1/CYP2B6*4 (7.3%) in Han subjects, whereas the most frequent genotypes in Uygur subjects were CYP2B6*1/CYP2B6*1 (36.3%), CYP2B6*1/CYP2B6*6 (25.3%), CYP2B6*1/CYP2B6*5 (5.5%) and CYP2B6*6/CYP2B6*6 (5.5%). The frequencies of 64C>T mutation in Han and Uygur Chinese were significantly lower than that in American Caucasian (P<0.05). These results indicate that there were marked ethnic differences in the mutant frequencies of CYP2B6 between Chinese and other ethnic groups. Further studies are warranted to explore the clinical impact of such ethnic differences.

Similar content being viewed by others
Abbreviations
- CI:
-
confidence interval
- PCR:
-
polymerase chain reaction
- RFLP:
-
restriction fragment length polymorphism
- SNP:
-
single nucleotide polymorphism
References
S. Rendic. Summary of information on human CYP enzymes: human P450 metabolism data. Drug Metab. Rev. 34:83–448 (2002).
S. Ekins, M. VandenBranden, B. J. Ring, and S. A. Wrighton. Examination of purported probes of human CYP2B6. Pharmacogenetics 7:165–179 (1997).
T. K. Chang, G. F. Weber, C. L. Crespi, and D. J. Waxman. Differential activation of cyclophosphamide and ifosphamide by cytochromes P-450 2B and 3A in human liver microsomes. Cancer Res. 53:5629–5637 (1993).
J. Wojcikowski, L. Pichard-Garcia, P. Maurel, and W. A. Daniel. Contribution of human cytochrome p-450 isoforms to the metabolism of the simplest phenothiazine neuroleptic promazine. Br. J. Pharmacol. 138:1465–1474 (2003).
J. G. Gerber, R. J. Rhodes, and J. Gal. Stereoselective metabolism of methadone N-demethylation by cytochrome P4502B6 and 2C19. Chirality 16:36–44 (2004).
Y. Oda and E. D. Kharasch. Metabolism of levo-alpha-Acetylmethadol (LAAM) by human liver cytochrome P450: involvement of CYP3A4 characterized by atypical kinetics with two binding sites. J. Pharmacol. Exp. Ther. 297:410–422 (2001).
H. Heyn, R. B. White, and J. C. Stevens. Catalytic role of cytochrome P4502B6 in the N-demethylation of S-mephenytoin. Drug Metab. Dispos. 24:948–954 (1996).
P. Riska, M. Lamson, T. MacGregor, J. Sabo, S. Hattox, J. Pav, and J. Keirns. Disposition and biotransformation of the antiretroviral drug nevirapine in humans. Drug Metab. Dispos. 27:895–901 (1999).
B. A. Ward, J. C. Gorski, D. R. Jones, S. D. Hall, D. A. Flockhart, and Z. Desta. The cytochrome P450 2B6 (CYP2B6) is the main catalyst of efavirenz primary and secondary metabolism: implication for HIV/AIDS therapy and utility of efavirenz as a substrate marker of CYP2B6 catalytic activity. J. Pharmacol. Exp. Ther. 306:287–300 (2003).
M. H. Court, S. X. Duan, L. M. Hesse, K. Venkatakrishnan, and D. J. Greenblatt. Cytochrome P-450 2B6 is responsible for interindividual variability of propofol hydroxylation by human liver microsomes. Anesthesiology 94:110–119 (2001).
S. R. Faucette, R. L. Hawke, E. L. Lecluyse, S. S. Shord, B. Yan, R. M. Laethem, and C. M. Lindley. Validation of bupropion hydroxylation as a selective marker of human cytochrome P450 2B6 catalytic activity. Drug Metab. Dispos. 28:1222–1230 (2000).
T. L. Domanski, K. M. Schultz, F. Roussel, J. C. Stevens, and J. R. Halpert. Structure–function analysis of human cytochrome P-450 2B6 using a novel substrate, site-directed mutagenesis, and molecular modeling. J. Pharmacol. Exp. Ther. 290:1141–1147 (1999).
T. J. Yang, K. W. Krausz, M. Shou, S. K. Yang, J. T. Buters, F. J. Gonzalez, and H. V. Gelboin. Inhibitory monoclonal antibody to human cytochrome P450 2B6. Biochem. Pharmacol. 55:1633–1640 (1998).
L. Gervot, B. Rochat, J. C. Gautier, F. Bohnenstengel, H. Kroemer, V. de Berardinis, H. Martin, P. Beaune, and I. de Waziers. Human CYP2B6: expression, inducibility and catalytic activities. Pharmacogenetics 9:295–306 (1999).
L. M. Hesse, P. He, S. Krishnaswamy, Q. Hao, K. Hogan, L. L. von Moltke, D. J. Greenblatt, and M. H. Court. Pharmacogenetic determinants of interindividual variability in bupropion hydroxylation by cytochrome P450 2B6 in human liver microsomes. Pharmacogenetics 14:225–238 (2004).
H. Kim, R. S. Wang, E. Elovaara, H. Raunio, O. Pelkonen, T. Aoyama, H. Vainio, and T. Nakajima. Cytochrome P450 isozymes responsible for the metabolism of toluene and styrene in human liver microsomes. Xenobiotica 27:657–665 (1997).
S. Ekins, M. Vandenbranden, B. J. Ring, J. S. Gillespie, T. J. Yang, H. V. Gelboin, and S. A. Wrighton. Further characterization of the expression in liver and catalytic activity of CYP2B6. J. Pharmacol. Exp. Ther. 286:1253–1259 (1998).
J. M. Rae, N. V. Soukhova, D. A. Flockhart, and Z. Desta. Triethylenethiophosphoramide is a specific inhibitor of cytochrome P450 2B6: implications for cyclophosphamide metabolism. Drug Metab. Dispos. 30:525–530 (2002).
T. Richter, M. Schwab, M. Eichelbaum, and U. M. Zanger. Inhibition of human CYP2B6 by N,N′,Ná-triethylenethiophosphoramide is irreversible and mechanism-based. Biochem. Pharmacol. 69:517–524 (2005).
U. M. Kent, D. E. Mills, R. V. Rajnarayanan, W. L. Alworth, and P. F. Hollenberg. Effect of 17-alpha-ethynylestradiol on activities of cytochrome P450 2B (P450 2B) enzymes: characterization of inactivation of P450s 2B1 and 2B6 and identification of metabolites. J. Pharmacol. Exp. Ther. 300:549–558 (2002).
T. Richter, T. E. Murdter, G. Heinkele, J. Pleiss, S. Tatzel, M. Schwab, M. Eichelbaum, and U. M. Zanger. Potent mechanism-based inhibition of human CYP2B6 by clopidogrel and ticlopidine. J. Pharmacol. Exp. Ther. 308:189–197 (2004).
I. Santisteban, S. Povey, E. A. Shephard, and I. R. Phillips. The major phenobarbital-inducible cytochrome P-450 gene subfamily (P450IIB) mapped to the long arm of human chromosome 19. Ann. Hum. Genet. 52(Pt 2):129–135 (1988).
T. Lang, K. Klein, J. Fischer, A. K. Nussler, P. Neuhaus, U. Hofmann, M. Eichelbaum, M. Schwab, and U. M. Zanger. Extensive genetic polymorphism in the human CYP2B6 gene with impact on expression and function in human liver. Pharmacogenetics 11:399–415 (2001).
V. Lamba, J. Lamba, K. Yasuda, S. Strom, J. Davila, M. L. Hancock, J. D. Fackenthal, P. K. Rogan, B. Ring, S. A. Wrighton, and E. G. Schuetz. Hepatic CYP2B6 expression: gender and ethnic differences and relationship to CYP2B6 genotype and CAR (constitutive androstane receptor) expression. J. Pharmacol. Exp. Ther. 307:906–922 (2003).
J. Zukunft, T. Lang, T. Richter, K. I. Hirsch-Ernst, A. K. Nussler, K. Klein, M. Schwab, M. Eichelbaum, and U. M. Zanger. A natural CYP2B6 TATA box polymorphism (-82T–>C) leading to enhanced transcription and relocation of the transcriptional start site. Mol. Pharmacol. 67:1772–1782 (2005).
K. Klein, T. Lang, T. Saussele, E. Barbosa-Sicard, W. H. Schunck, M. Eichelbaum, M. Schwab, and U. M. Zanger. Genetic variability of CYP2B6 in populations of African and Asian origin: allele frequencies, novel functional variants, and possible implications for anti-HIV therapy with efavirenz. Pharmacogenet. Genomics 15:861–873 (2005).
J. Wang, A. Sonnerborg, A. Rane, F. Josephson, S. Lundgren, L. Stahle, and M. Ingelman-Sundberg. Identification of a novel specific CYP2B6 allele in Africans causing impaired metabolism of the HIV drug efavirenz. Pharmacogenet. Genomics 16:191–198 (2006).
H. Jinno, T. Tanaka-Kagawa, A. Ohno, Y. Makino, E. Matsushima, N. Hanioka, and M. Ando. Functional characterization of cytochrome P450 2B6 allelic variants. Drug Metab. Dispos. 31:398–403 (2003).
N. Ariyoshi, M. Miyazaki, K. Toide, Y. Sawamura, and T. Kamataki. A single nucleotide polymorphism of CYP2b6 found in Japanese enhances catalytic activity by autoactivation. Biochem. Biophys. Res. Commun. 281:1256–1260 (2001).
S. Guan, M. Huang, E. Chan, X. Chen, W. Duan, and S.F. Zhou. Genetic polymorphisms of cytochrome P450 2B6 gene in Han Chinese. Eur J Pharm Sci. (in press)
S. Yamano, P. T. Nhamburo, T. Aoyama, U. A. Meyer, T. Inaba, W. Kalow, H. V. Gelboin, O. W. McBride, and F. J. Gonzalez. cDNA cloning and sequence and cDNA-directed expression of human P450 IIB1: identification of a normal and two variant cDNAs derived from the CYP2B locus on chromosome 19 and differential expression of the IIB mRNAs in human liver. Biochemistry 28:7340–7348 (1989).
P. Chomczynski and N. Sacchi. Single-step method of RNA isolation by acid guanidinium thiocyanate–phenol–chloroform extraction. Anal. Biochem. 162:156–159 (1987).
R. G. Newcombe. Two-sided confidence intervals for the single proportion: comparison of seven methods. Stat. Med. 17:857–872 (1998).
M. Hiratsuka, Y. Takekuma, N. Endo, K. Narahara, S. I. Hamdy, Y. Kishikawa, M. Matsuura, Y. Agatsuma, T. Inoue, and M. Mizugaki. Allele and genotype frequencies of CYP2B6 and CYP3A5 in the Japanese population. Eur. J. Clin. Pharmacol. 58:417–421 (2002).
D. A. Evans, H. L. McLeod, S. Pritchard, M. Tariq, and A. Mobarek. Interethnic variability in human drug responses. Drug Metab. Dispos. 29:606–610 (2001).
H. G. Xie, R. B. Kim, A. J. J. Wood, and C. M. Stein. Molecular basis of ethnic differences in drug disposition and response. Annu. Rev. Pharmacol. Toxicol. 41:815–850 (2001).
B. Chowbay, S. Zhou, and E. J. Lee. An interethnic comparison of polymorphisms of the genes encoding drug-metabolizing enzymes and drug transporters: experience in Singapore. Drug Metab. Rev. 37:327–378 (2005).
J. Y. Cho, H. S. Lim, J. Y. Chung, K. S. Yu, J. R. Kim, S. G. Shin, and I. J. Jang. Haplotype structure and allele frequencies of CYP2B6 in a Korean population. Drug Metab. Dispos. 32:1341–1344 (2004).
D. Burger, I. van der Heiden, C. la Porte, M. van der Ende, P. Groeneveld, C. Richter, P. Koopmans, F. Kroon, H. Sprenger, J. Lindemans, P. Schenk, and R. van Schaik. Interpatient variability in the pharmacokinetics of the HIV non-nucleoside reverse transcriptase inhibitor efavirenz: the effect of gender, race, and CYP2B6 polymorphism. Br. J. Clin. Pharmacol. 61:148–154 (2006).
D. W. Haas, L. M. Smeaton, R. W. Shafer, G. K. Robbins, G. D. Morse, L. Labbe, G. R. Wilkinson, D. B. Clifford, R. T. D'Aquila, V. De Gruttola, R. B. Pollard, T. C. Merigan, M. S. Hirsch, A. L. George Jr., J. P. Donahue, and R. B. Kim. Pharmacogenetics of long-term responses to antiretroviral regimens containing efavirenz and/or nelfinavir: an Adult AIDS Clinical Trials Group Study. J. Infect. Dis. 192:1931–1942 (2005).
M. Rotger, S. Colombo, H. Furrer, G. Bleiber, T. Buclin, B. L. Lee, O. Keiser, J. Biollaz, L. Decosterd, and A. Telenti. Influence of CYP2B6 polymorphism on plasma and intracellular concentrations and toxicity of efavirenz and nevirapine in HIV-infected patients. Pharmacogenet. Genomics. 15:1–5 (2005).
S. Rodriguez-Novoa, P. Barreiro, A. Rendon, I. Jimenez-Nacher, J. Gonzalez-Lahoz, and V. Soriano. Influence of 516G>T polymorphisms at the gene encoding the CYP450-2B6 isoenzyme on efavirenz plasma concentrations in HIV-infected subjects. Clin. Infect. Dis. 40:1358–1361 (2005).
D. W. Haas, H. J. Ribaudo, R. B. Kim, C. Tierney, G. R. Wilkinson, R. M. Gulick, D. B. Clifford, T. Hulgan, C. Marzolini, and E. P. Acosta. Pharmacogenetics of efavirenz and central nervous system side effects: an Adult AIDS Clinical Trials Group study. Aids 18:2391–2400 (2004).
S. M. Yule, A. V. Boddy, M. Cole, L. Price, R. Wyllie, M. J. Tasso, A. D. Pearson, and J. R. Idle. Cyclophosphamide pharmacokinetics in children. Br. J. Clin. Pharmacol. 41:13–19 (1996).
A. V. Boddy, Y. Furtun, S. Sardas, O. Sardas, and J. R. Idle. Individual variation in the activation and inactivation of metabolic pathways of cyclophosphamide. J. Natl. Cancer. Inst. 84:1744–1748 (1992).
T. L. Chen, J. L. Passos-Coelho, D. A. Noe, M. J. Kennedy, K. C. Black, O. M. Colvin, and L. B. Grochow. Nonlinear pharmacokinetics of cyclophosphamide in patients with metastatic breast cancer receiving high-dose chemotherapy followed by autologous bone marrow transplantation. Cancer Res. 55:810–816 (1995).
R. Schmidt, F. Baumann, H. Hanschmann, F. Geissler, and R. Preiss. Gender difference in ifosfamide metabolism by human liver microsomes. Eur. J. Drug Metab. Pharmacokinet. 26:193–200 (2001).
H. Wang and M. Negishi. Transcriptional regulation of cytochrome p450 2B genes by nuclear receptors. Curr. Drug Metab. 4:515–525 (2003).
T. Lang, K. Klein, T. Richter, A. Zibat, R. Kerb, M. Eichelbaum, M. Schwab, and U. M. Zanger. Multiple novel nonsynonymous CYP2B6 gene polymorphisms in Caucasians: demonstration of phenotypic null alleles. J. Pharmacol. Exp. Ther. 311:34–43 (2004).
M. Spatzenegger, H. Liu, Q. Wang, A. Debarber, D. R. Koop, and J. R. Halpert. Analysis of differential substrate selectivities of CYP2B6 and CYP2E1 by site-directed mutagenesis and molecular modeling. J. Pharmacol. Exp. Ther. 304:477–487 (2003).
Acknowledgments
The authors thank Prof. Zanger, Dr. Kathrin Klein (Dr.Margarete Fischer-Bosch Institut for Klinische Pharmakologie, Stuttgart, Germany), and Prof. Mizugaki, Dr. Masahiro Hiratsuka (Department of Clinical Pharmaceutics Tohoku Pharmaceutical University, Japan) for providing DNA reference samples. This work was supported by the National Nature Science Fund of China (No. 30572231), Guangdong Nature Science Fund (no. 2003: 36622) and the National University of Singapore Academic Research Funds (nos. R-148-000-047-101 and R-148-000-067-112).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Guan, S., Huang, M., Li, X. et al. Intra- and Inter-ethnic Differences in the Allele Frequencies of Cytochrome P450 2B6 Gene in Chinese. Pharm Res 23, 1983–1990 (2006). https://doi.org/10.1007/s11095-006-9083-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-006-9083-5