ABSTRACT
Purpose
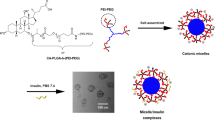
Pancreatic polypeptide (PP) has important glucoregulatory functions and thereby holds significance in the treatment of diabetes and obesity. However, short plasma half-life and aggregation propensity of PP in aqueous solution, limits its therapeutic application. To address these issues, we prepared and characterized a formulation of PP in sterically stabilized micelles (SSM) that protects and stabilizes PP in its active conformation.
Methods
PP-SSM was prepared by incubating PP with SSM dispersion in buffer. Peptide-micelle association and freeze-drying efficacy of the formulation was characterized in phosphate buffers with or without sodium chloride using dynamic light scattering, fluorescence spectroscopy and circular dichroism. The degradation kinetics of PP-SSM in presence of proteolytic enzyme was determined using HPLC and bioactivity of the formulation was evaluated by in vitro cAMP inhibition study.
Results
PP self-associated with SSM and this interaction was influenced by presence/absence of sodium chloride in the buffer. The formulation was effectively lyophilized, demonstrating feasibility for its long-term storage. The stability of peptide against proteolytic degradation was significantly improved and PP in SSM retained its bioactivity in vitro.
Conclusions
Self-association of PP with phospholipid micelles addressed the delivery issues of the peptide. This nanomedicine should be further developed for the treatment of diabetes.











Similar content being viewed by others
Abbreviations
- ANOVA:
-
analysis of variance
- cAMP:
-
cyclic adenosine monophosphate
- CD:
-
circular dichroism
- CMC:
-
critical micellar concentration
- CP:
-
chronic pancreatitis
- DLS:
-
dynamic light scattering
- DSPE-PEG2000 :
-
distearoyl phosphatidylethanolamine-polyethylene glycol2000
- EMEM:
-
eagle’s minimum essential media
- FD:
-
freeze-drying
- IBMX:
-
3-Isobutyl-1-methylxanthine
- NPY:
-
neuropeptide Y
- NS:
-
normal saline
- PB:
-
phosphate buffer
- PBS:
-
phosphate buffered saline
- PD:
-
pancreatogenic diabetes
- PP:
-
pancreatic polypeptide
- RP-HPLC:
-
peversed phase high pressure liquid chromatography
- SD:
-
standard deviation
- SSM:
-
sterically stabilized micelles
References
Lonovics J, Devitt P, Watson LC, Thompson JC. Pancreatic polypeptide. A review. Arch Surg. 1981;116(10):1256–64.
Schmidt PT, Naslund E, Gryback P, Jacobsson H, Holst JJ, Hilsted L, et al. A role for pancreatic polypeptide in the regulation of gastric emptying and short-term metabolic control. J Clin Endocrinol Metab. 2005;90(9):5241–6.
Langlois A, Corring T, Levenez F, Cuber JC, Chayvialle JA. Effects of pancreatic polypeptide on biliary flow and bile acid secretion stimulated by secretin and cholecystokinin in the conscious pig. Regul Pept. 1990;27(1):139–47.
Lerch M, Kamimori H, Folkers G, Aguilar MI, Beck-Sickinger AG, Zerbe O. Strongly altered receptor binding properties in PP and NPY chimeras are accompanied by changes in structure and membrane binding. Biochemistry. 2005;44(25):9255–64.
Bard JA, Walker MW, Branchek TA, Weinshank RL. Cloning and functional expression of a human Y4 subtype receptor for pancreatic polypeptide, neuropeptide Y, and peptide YY. J Biol Chem. 1995;270(45):26762–5.
Seymour NE, Spector SA, Andersen DK, Elm MS, Whitcomb DC. Overexpression of hepatic pancreatic polypeptide receptors in chronic pancreatitis. J Surg Res. 1998;76(1):47–52.
Hennig R, Kekis PB, Friess H, Adrian TE, Buchler MW. Pancreatic polypeptide in pancreatitis. Peptides. 2002;23(2):331–8.
Adrian TE, Besterman HS, Mallinson CN, Garalotis C, Bloom SR. Impaired pancreatic polypeptide release in chronic pancreatitis with steatorrhoea. Gut. 1979;20(2):98–101.
Cui Y, Andersen DK. Pancreatogenic diabetes: special considerations for management. Pancreatology. 2011;11(3):279–94.
Seymour NE, Andersen DK. Pancreatic polypeptide and glucose metabolism. In: Jr G, editor. Gastrointestinal endocrinology. New Jersey: Humana; 1999. p. 321–34.
Seymour NE, Volpert AR, Andersen DK. Regulation of hepatic insulin receptors by pancreatic polypeptide in fasting and feeding. J Surg Res. 1996;65(1):1–4.
Seymour NE, Volpert AR, Lee EL, Andersen DK, Hernandez C. Alterations in hepatocyte insulin binding in chronic pancreatitis: effects of pancreatic polypeptide. Am J Surg. 1995;169(1):105–9.
Asakawa A, Inui A, Yuzuriha H, Ueno N, Katsuura G, Fujimiya M, et al. Characterization of the effects of pancreatic polypeptide in the regulation of energy balance. Gastroenterology. 2003;124(5):1325–36.
Steppan CM, Lazar MA. Resistin and obesity-associated insulin resistance. Trends Endocrinol Metab. 2002;13(1):18–23.
Adrych K, Smoczynski M, Sledzinski T, Dettlaff-Pokora A, Goyke E, Swierczynski J. Increased serum resistin concentration in patients with chronic pancreatitis: possible cause of pancreatic fibrosis. J Clin Gastroenterol. 2009;43(1):63–8.
Andersen DK. Mechanisms and emerging treatments of the metabolic complications of chronic pancreatitis. Pancreas. 2007;35(1):1–15.
Brunicardi FC, Chaiken RL, Ryan AS, Seymour NE, Hoffmann JA, Lebovitz HE, et al. Pancreatic polypeptide administration improves abnormal glucose metabolism in patients with chronic pancreatitis. J Clin Endocrinol Metab. 1996;81(10):3566–72.
Sun YS, Brunicardi FC, Druck P, Walfisch S, Berlin SA, Chance RE, et al. Reversal of abnormal glucose metabolism in chronic pancreatitis by administration of pancreatic polypeptide. Am J Surg. 1986;151(1):130–40.
Seymour NE, Brunicardi FC, Chaiken RL, Lebovitz HE, Chance RE, Gingerich RL, et al. Reversal of abnormal glucose production after pancreatic resection by pancreatic polypeptide administration in man. Surgery. 1988;104(2):119–29.
Adrian TE, Greenberg GR, Besterman HS, Bloom SR. Pharmacokinetics of pancreatic polypeptide in man. Gut. 1978;19(10):907–9.
Baxter J, Minnion J, Shilto-Cuenco J, Tan T, Murphy K, Ghatei M, et al. Pancreatic polypeptide: a novel substrate for the endopeptidase neprilysin. Endocr Abstr. 2010;21:P133.
Swierczek JS, Pawlik W, Konturek SJ, Gustaw P, Dobrzanska M, Bielanski W, et al. Organ removal and disappearance half-time of synthetic human pancreatic polypeptide. Digestion. 1982;25(3):197–200.
Ashok B, Arleth L, Hjelm RP, Rubinstein I, Onyuksel H. In vitro characterization of PEGylated phospholipid micelles for improved drug solubilization: effects of PEG chain length and PC incorporation. J Pharm Sci. 2004;93(10):2476–87.
Krishnadas A, Rubinstein I, Onyuksel H. Sterically stabilized phospholipid mixed micelles: in vitro evaluation as a novel carrier for water-insoluble drugs. Pharm Res. 2003;20(2):297–302.
Onyuksel H, Jeon E, Rubinstein I. Nanomicellar paclitaxel increases cytotoxicity of multidrug resistant breast cancer cells. Cancer Lett. 2009;274(2):327–30.
Onyuksel H, Sejourne F, Suzuki H, Rubinstein I. Human VIP-alpha: a long-acting, biocompatible and biodegradable peptide nanomedicine for essential hypertension. Peptides. 2006;27(9):2271–5.
Lim SB, Rubinstein I, Sadikot RT, Artwohl JE, Onyuksel H. A novel peptide nanomedicine against acute lung injury: GLP-1 in phospholipid micelles. Pharm Res. 2011;28(3):662–72.
Koo OM, Rubinstein I, Onyuksel H. Actively targeted low-dose camptothecin as a safe, long-acting, disease-modifying nanomedicine for rheumatoid arthritis. Pharm Res. 2011;28(4):776–87.
Koo OM, Rubinstein I, Onyuksel H. Camptothecin in sterically stabilized phospholipid micelles: a novel nanomedicine. Nanomedicine: Nanomed-Nanotechnol Biol Med. 2005;1(1):77–84.
Kuzmis A, Lim SB, Desai E, Jeon E, Lee BS, Rubinstein I, et al. Micellar nanomedicine of human neuropeptide Y. Nanomedicine. 2011;7(4):464–71.
Vukovic L, Khatib FA, Drake SP, Madriaga A, Brandenburg KS, Kral P, et al. Structure and dynamics of highly PEG-ylated sterically stabilized micelles in aqueous media. J Am Chem Soc. 2011;133(34):13481–8.
Kanazawa I, Hamaguchi K. Unfolding by temperature and guanidine hydrochloride of chicken pancreatic polypeptide. J Biochem. 1986;100(1):207–12.
Glover ID, Barlow DJ, Pitts JE, Wood SP, Tickle IJ, Blundell TL, et al. Conformational studies on the pancreatic polypeptide hormone family. Eur J Biochem. 1984;142(2):379–85.
Lim SB, Rubinstein I, Önyüksel H. Freeze drying of peptide drugs self-associated with long-circulating, biocompatible and biodegradable sterically stabilized phospholipid nanomicelles. Int J Pharm. 2008;356(1–2):345–50.
Taylor TC, Thompson DO, Ebner KE, Kimmel JR, Rawitch AB. An immunochemical study of avian pancreatic polypeptide: the nature of the principle epitope. Mol Immunol. 1988;25(10):961–73.
Li A, Ritter S. Functional expression of neuropeptide Y receptors in human neuroblastoma cells. Regul Pept. 2005;129(1–3):119–24.
Lazo ND, Downing DT. Stabilization of amphipathic alpha-helical and beta-helical conformations in synthetic peptides in the presence and absence of ionic interactions. J Pept Res. 1998;51(1):85–9.
Lerch M, Gafner V, Bader R, Christen B, Folkers G, Zerbe O. Bovine Pancreatic Polypeptide (bPP) undergoes significant changes in conformation and dynamics upon binding to DPC micelles. J Mol Biol. 2002;322(5):1117–33.
Tonan K, Kawata Y, Hamaguchi K. Conformations of isolated fragments of pancreatic polypeptide. Biochemistry. 1990;29(18):4424–9.
ACKNOWLEDGMENTS & DISCLOSURES
The authors thank Dr. Bao-Shiang Lee for synthesizing PP used in the research.
The study was supported in part by NIH grant CA121797 and UIC university scholar award.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Banerjee, A., Onyuksel, H. Human Pancreatic Polypeptide in a Phospholipid-Based Micellar Formulation. Pharm Res 29, 1698–1711 (2012). https://doi.org/10.1007/s11095-012-0718-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-012-0718-4