Abstract
Purpose. To investigate in vitro if P-glycoprotein (P-gp) transport can differentiate between antibiotic drugs exhibiting increased active renal clearance (CLr) in cystic fibrosis (CF) patients (i.e., dicloxacillin, trimethoprim) and drugs that do not exhibit this phenomenon (i.e., cefsulodin, sulfamethoxazole).
Methods. Transport studies were carried out in MDCK (wild type) and MDR1-MDCK (P-gp overexpressing) cells that were grown to confluence on Transwell inserts. [14C]-mannitol transport and transepithelial electrical resistance (TEER) were measured to validate the integrity of the cells. Drug concentrations were analyzed using HPLC.
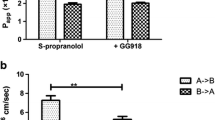
Results. Dicloxacillin and trimethoprim are substrates of P-gp (B→A/A→B ratios in MDR1-MDCK cells are 32 and 50, respectively). P-gp inhibitors (i.e., GG918, cyclosporine, ketoconazole, vinblastine) decreased the B→A transport of dicloxacillin and trimethoprim and increased the A→B transport of trimethoprim while non-P-gp inhibitors (e.g., PAH) had no effect. In contrast, cefsulodin and sulfamethoxazole are not substrates of P-gp (B→A/A→B values in MDCK and MDR1-MDCK cells are ∼1).
Conclusions. Our in vitro studies suggest that P-glycoprotein may play a role in increasing renal clearance of drug substrates in CF patients. Dicloxacillin and trimethoprim, which are both substrates of P-gp, show increased active renal clearance in CF patients while cefsulodin and sulfamethoxazole, which are not P-gp substrates, do not show increased active renal clearance in CF patients.
Similar content being viewed by others
REFERENCES
M. Welsh, L. Tsui, T. Boat, and A. Beaudeut. Cystic Fibrosis. In C. Scriver, A. Beaudet, W. Sly, and V. David (eds.), The Metabolic and Molecular Bases of Inherited Disease, McGraw-Hill, New York, 1995 pp. 3799–3876.
W. J. Jusko, L. L. Mosovich, L. M. Gerbracht, M. E. Mattar, and S. J. Yaffe. Enhanced renal excretion of dicloxacillin in patients with cystic fibrosis. Pediatrics 56:1038–1044 (1975).
S. J. Yaffe, L. M. Gerbracht, L. L. Mosovich, M. E. Mattar, M. Danish, and W. J. Jusko. Pharmacokinetics of methicillin in patients with cystic fibrosis. J. Infect. Dis. 135:828–831 (1977).
M. Spino, R. P. Chai, A. F. Isles, J. J. Thiessen, A. Tesoro, R. Gold, and S. M. MacLeod. Cloxacillin absorption and disposition in cystic fibrosis. J. Pediatr. 105:829–835 (1984).
R. M. Hutabarat, J. D. Unadkat, C. Sahajwalla, S. McNamara, B. Ramsey, and A. L. Smith. Disposition of drugs in cystic fibrosis. I. Sulfamethoxazole and trimethoprim. Clin. Pharmacol. Ther. 49:402–409 (1991).
R. de Groot, B. D. Hack, A. Weber, D. Chaffin, B. Ramsey, and A. L. Smith. Pharmacokinetics of ticarcillin in patients with cystic fibrosis: a controlled prospective study. Clin. Pharmacol. Ther. 47:73–78 (1990).
J. P. Wang, J. D. Unadkat, S. M. al-Habet, T. A. O'Sullivan, J. Williams-Warren, A. L. Smith, and B. Ramsey. Disposition of drugs in cystic fibrosis. IV. Mechanisms for enhanced renal clearance of ticarcillin. Clin. Pharmacol. Ther. 54:293–302 (1993).
R. de Groot and A. L. Smith. Antibiotic pharmacokinetics in cystic fibrosis. Differences and clinical significance. Clin. Pharmacokinet. 13:228–253 (1987).
J. Prandota. Clinical pharmacology of antibiotics and other drugs in cystic fibrosis. Drugs 35:542–578 (1988).
E. Rey, J. M. Treluyer, and G. Pons. Drug disposition in cystic fibrosis. Clin. Pharmacokinet. 35:313–329 (1998).
F. Sörgel, U. Stephan, H. G. Wiesemann, B. Gottschalk, C. Stehr, M. Rey, H. B. Bowing, H. C. Dominick, and M. Geldmacher von Mallinckrodt. High dose treatment with antibiotics in cystic fibrosis—a reappraisal with special reference to the pharmacokinetics of beta-lactams and new fluoroquinolones in adult CF-patients. Infection 15:385–396 (1987).
M. Spino. Pharmacokinetics of drugs in cystic fibrosis. Clin. Rev. Allergy 9:169–210 (1991).
D. J. Touw. Clinical pharmacokinetics of antimicrobial drugs in cystic fibrosis. Pharm. World Sci. 20:149–160 (1998).
A. Arvidsson, G. Alvan, and B. Strandvik. Difference in renal handling of cefsulodin between patients with cystic fibrosis and normal subjects. Acta Paediatr. Scand. 72:293–294 (1983).
J. S. Leeder, M. Spino, A. F. Isles, A. M. Tesoro, R. Gold, and S. M. MacLeod. Ceftazidime disposition in acute and stable cystic fibrosis. Clin. Pharmacol. Ther. 36:355–362 (1984).
J. Levy, A. L. Smith, J. R. Koup, J. Williams-Warren, and B. Ramsey. Disposition of tobramycin in patients with cystic fibrosis: a prospective controlled study. J. Pediatr. 105:117–124 (1984).
M. Spino, R. P. Chai, A. F. Isles, J. W. Balfe, R. G. Brown, J. J. Thiessen, and S. M. MacLeod. Assessment of glomerular filtration rate and effective renal plasma flow in cystic fibrosis. J. Pediatr. 107:64–70 (1985).
U. Berg, E. Kusoffsky, and B. Strandvik. Renal function in cystic fibrosis with special reference to the renal sodium handling. Acta Paediatr. Scand. 71:833–838 (1982).
A. Hedman, G. Alvan, B. Strandvik, and A. Arvidsson. Increased renal clearance of cefsulodin due to higher glomerular filtration rate in cystic fibrosis. Clin. Pharmacokinet. 18:168–175 (1990).
B. Strandvik, U. Berg, A. Kallner, and E. Kusoffsky. Effect on renal function of essential fatty acid supplementation in cystic fibrosis. J. Pediatr. 115:242–250 (1989).
W. Breuer, I. N. Slotki, D. A. Ausiello, and I. Z. Cabantchik. Induction of multidrug resistance downregulates the expression of CFTR in colon epithelial cells. Am. J. Physiol. 265:C1711–C1715 (1993).
A. E. Trezise, P. R. Romano, D. R. Gill, S. C. Hyde, F. V. Sepulveda, M. Buchwald, and C. F. Higgins. The multidrug resistance and cystic fibrosis genes have complementary patterns of epithelial expression. EMBO J. 11:4291–4303 (1992).
A. E. Trezise, R. Ratcliff, T. E. Hawkins, M. J. Evans, T. C. Freeman, P. R. Romano, C. F. Higgins, and W. H. Colledge. Co-ordinate regulation of the cystic fibrosis and multidrug resistance genes in cystic fibrosis knockout mice. Hum. Mol. Genet. 6:527–537 (1997).
S. Bremer, T. Hoof, M. Wilke, R. Busche, B. Scholte, J. R. Riordan, G. Maass, and B. Tummler. Quantitative expression patterns of multidrug-resistance P-glycoprotein (MDR1) and differentially spliced cystic-fibrosis transmembrane-conductance regulator mRNA transcripts in human epithelia. Eur. J. Biochem. 206:137–149 (1992).
C. G. Vanoye, A. F. Castro, T. Pourcher, L. Reuss, and G. A. Altenberg. Phosphorylation of P-glycoprotein by PKA and PKC modulates swelling-activated Cl-currents. Am. J. Physiol. 276:C370–C378 (1999).
I. Pastan, M. M. Gottesman, K. Ueda, E. Lovelace, A. V. Rutherford, and M. C. Willingham. A retrovirus carrying an MDR1 cDNA confers multidrug resistance and polarized expression of P-glycoprotein in MDCK cells. Proc. Natl. Acad. Sci. USA 85:4486–4490 (1988).
Y. Zhang and L. Z. Benet. Characterization of P-glycoprotein mediated transport of K02, a novel vinylsulfone peptidomimetic cysteine protease inhibitor, across MDR1-MDCK and Caco-2 cell monolayers. Pharm. Res. 15:1520–1524 (1998).
F. Jehl, P. Birckel, and H. Monteil. Hospital routine analysis of penicillins. Third-generation cephalosporins and aztreonam by conventional and high-speed high-performance liquid chromatography. J. Chromatogr. 413:109–119 (1987).
M. Susanto and L. Z. Benet. Investigating the role of P-glycoprotein on drugs exhibiting enhanced renal clearance in cystic fibrosis patients. In:Proceedings of the Millenial World Congress of Pharmaceutical Sciences, San Francisco, California USA, 16-20 April 2000, pp. 94.
J. W. Polli, S. A. Wring, J. E. Humphreys, L. Huang, J. B. Morgan, L. O. Webster, and C. S. Serabjit-Singh. Rational use of in vitro p-glycoprotein assays in drug discovery. J. Pharmacol. Exp. Ther. 299:620–628 (2001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Susanto, M., Benet, L.Z. Can the Enhanced Renal Clearance of Antibiotics in Cystic Fibrosis Patients Be Explained by P-Glycoprotein Transport?. Pharm Res 19, 457–462 (2002). https://doi.org/10.1023/A:1015191511817
Issue Date:
DOI: https://doi.org/10.1023/A:1015191511817