Abstract
Background: Liver cirrhosis is a progressive disease characterized by loss of functional hepatocytes with concomitant connective tissue and nodule formation in the liver. The morphological and physiological changes associated with the disease substantially affect drug pharmacokinetics. Whole-body physiologically based pharmacokinetic (WB-PBPK) modelling is a predictive technique that quantitatively relates the pharmacokinetic parameters of a drug to such (patho-)physiological conditions.
Objective: To extend an existing WB-PBPK model, based on the physiological changes associated with liver cirrhosis, which allows for prediction of drug pharmacokinetics in patients with liver cirrhosis.
Methods: The literature was searched for quantitative measures of the physiological changes associated with the presence of Child-Pugh class A through C liver cirrhosis. The parameters that were included were the organ blood flows, cardiac index, plasma binding protein concentrations, haematocrit, functional liver volume, hepatic enzymatic activity and glomerular filtration rate. Predictions of pharmacokinetic profiles and parameters were compared with literature data for the model compounds alfentanil, lidocaine (lignocaine), theophylline and levetiracetam.
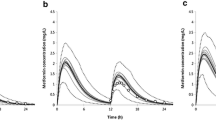
Results: The predicted versus observed plasma concentration-time profiles for alfentanil and lidocaine were similar, such that the pharmacokinetic changes associated with Child-Pugh class A, B and C liver cirrhosis were adequately described. The theophylline elimination half-life was greatly increased in Child-Pugh class B and C patients compared with controls, as predicted by the model. Levetiracetam urinary excretion was consistently reduced with disease progression and very closely resembled observed values.
Conclusion: Consideration of the physiological differences between healthy individuals and patients with liver cirrhosis was important for the simulation of drug pharmacokinetics in this compromised group. The WB-PBPK model was altered to incorporate these physiological differences with the result of adequate simulation of drug pharmacokinetics. The information provided in this study will allow other researchers to further validate this liver cirrhosis model within a WB-PBPK model.








Similar content being viewed by others
References:
Bosch J, Garcia-Pagan JC. Complications of cirrhosis: I. Portal hypertension. J Hepatol 2000; 32 (1 Suppl.): 141–56
Schuppan D, Afdhal NH. Liver cirrhosis. Lancet 2008; 371(9615): 838–51
Dincer D, Besisk F, Demirkol O, et al. Relationships between hemodynamic alterations and Child-Pugh score in patients with cirrhosis. Hepatogastroenterology 2005; 52(65): 1521–5
Andersen ME. Toxicokinetic modeling and its applications in chemical risk assessment. Toxicol Lett 2003; 138(1-2): 9–27
Edginton AN, Schmitt W, Willmann S. Development and evaluation of a generic physiologically based pharmacokinetic model for children. Clin Pharmacokinet 2006; 45(10): 1013–34
Bjorkman S, Wada DR, Berling BM, et al. Prediction of the disposition of midazolam in surgical patients by a physiologically based pharmacokinetic model. J Pharm Sci 2001; 90(9): 1226–41
Lupfert C, Reichel A. Development and application of physiologically based pharmacokinetic-modeling tools to support drug discovery. Chem Biodivers 2005; 2(11): 1462–86
Willmann S, Schmitt W, Keldenich J, et al. A physiological model for the estimation of the fraction dose absorbed in humans. J Med Chem 2004; 47(15): 4022–31
International Commission on Radiological Protection (ICRP). Basic anatomical and physiological data for use in radiological protection: reference values. ICRP publication no. 89. Amsterdam: Elsevier Science, 2002
Bjorkman S. Prediction of drug disposition in infants and children by means of physiologically based pharmacokinetic (PBPK) modelling: theophylline and midazolam as model drugs. Br J Clin Pharmacol 2005; 59(6): 691–704
Annet L, Materne R, Danse E, et al. Hepatic flow parameters measured with MR imaging and Doppler US: correlations with degree of cirrhosis and portal hypertension. Radiology 2003; 229(2): 409–14
Yamamoto M, Iwasa M, Matsumura K, et al. Improvement of regional cerebral blood flow after oral intake of branched-chain amino acids in patients with cirrhosis. World J Gastroenterol 2005; 11(43): 6792–9
Chawla Y, Santa N, Dhiman RK, et al. Portal hemodynamics by duplex Doppler sonography in different grades of cirrhosis. Dig Dis Sci 1998; 43(2): 354–7
Froomes PR, Morgan DJ, Smallwood RA, et al. Comparative effects of oxygen supplementation on theophylline and acetaminophen clearance in human cirrhosis. Gastroenterology 1999; 116(4): 915–20
Woitas RP, Stoffel-Wagner B, Flommersfeld S, et al. Correlation of serum concentrations of cystatin C and creatinine to inulin clearance in liver cirrhosis. Clin Chem 2000; 46(5): 712–5
Barry M, Keeling PW, Weir D, et al. Severity of cirrhosis and the relationship of alpha 1-acid glycoprotein concentration to plasma protein binding of lidocaine. Clin Pharmacol Ther 1990; 47(3): 366–70
Wong F, Girgrah N, Graba J, et al. The cardiac response to exercise in cirrhosis. Gut 2001; 49(2): 268–75
Sansoe G, Ferrari A, Castellana CN, et al. Cimetidine administration and tubular creatinine secretion in patients with compensated cirrhosis. Clin Sci (Lond) 2002; 102(1): 91–8
Proulx NL, Akbari A, Garg AX, et al. Measured creatinine clearance from timed urine collections substantially overestimates glomerular filtration rate in patients with liver cirrhosis: a systematic review and individual patient meta-analysis. Nephrol Dial Transplant 2005; 20(8): 1617–22
Willmann S, Lippert J, Schmitt W. From physicochemistry to absorption and distribution: predictive mechanistic modelling and computational tools. Expert Opin Drug Meta Toxicol 2005; 1(1): 159–68
Virgolini I, Muller C, Angelberger P, et al. Functional liver imaging with 99Tcm-galactosyl-neoglycoalbumin (NGA) in alcoholic liver cirrhosis and liver fibrosis. Nucl Med Commun 1991; 12(6): 507–17
George J, Murray M, Byth K, et al. Differential alterations of cytochrome P450 proteins in livers from patients with severe chronic liver disease. Hepatology 1995; 21(1): 120–8
Hardman JG, Limbird LE, Gilman A. Goodman and Gilman’s: the pharmacological basis of therapeutics. 10th ed. New York: McGraw Hill, 2001
Brockmöller J, Thomsen T, Wittstock M, et al. Pharmacokinetics of levetiracetam in patients with moderate to severe liver cirrhosis (Child-Pugh classes A, B, and C): characterization by dynamic liver function tests. Clin Pharmacol Ther 2005; 77(6): 529–41
Coupez R, Nicolas JM, Browne TR. Levetiracetam, a new antiepileptic agent: lack of in vitro and in vivo pharmacokinetic interaction with valproic acid. Epilepsia 2003; 44(2): 171–8
Willmann S, Lippert J, Sevestre M, et al. PK-Sim®: a physiologically based pharmacokinetic ‘whole-body’ model. Biosilico 2003; 1(4): 121–4
Willmann S, Hohn K, Edginton A, et al. Development of a physiology-based whole-body population model for assessing the influence of individual variability on the pharmacokinetics of drugs. J Pharmacokinet Pharmacodyn 2007; 34(3): 401–31
US National Center for Health Statistics. Third National Health and Nutrition Examination Survey (NHANES III). Hyattsville (MD): National Center for Health Statistics Hyattsville, 1997 [online]. Available from URL: http://www.cdc.gov/nchs/datawh/nchsdefs/bmi.htm [Accessed 2008 Aug 19]
Sheiner LB, Beal SL. Some suggestions for measuring predictive performance. J Pharmacokinet Biopharma 1981; 9(4): 503–12
Roure P, Jean N, Leclerc AC, et al. Pharmacokinetics of alfentanil in children undergoing surgery. Br J Anaesth 1987; 59(11): 1437–40
Macfie AG, Magides AD, Reilly CS. Disposition of alfentanil in burns patients. Br J Anaesth 1992; 69(5): 447–50
Chauvin M, Bonnet F, Montembault C, et al. The influence of hepatic plasma flow on alfentanil plasma concentration plateaus achieved with an infusion model in humans: measurement of alfentanil hepatic extraction coefficient. Anesth Analg 1986; 65(10): 999–1003
Kharasch ED, Russell M, Mautz D, et al. The role of cytochrome P450 3A4 in alfentanil clearance: implications for interindividual variability in disposition and perioperative drug interactions. Anesthesiology 1997; 87(1): 36–50
Mazoit JX, Dalens BJ. Pharmacokinetics of local anaesthetics in infants and children. Clin Pharmacokinet 2004; 43(1): 17–32
Bailey DN, Briggs JR. The binding of selected therapeutic drugs to human serum [alpha]-1 acid glycoprotein and to human serum albumin in vitro. Ther Drug Monit 2004; 26(1): 40–3
Bertz RJ, Granneman GR. Use of in vitro and in vivo data to estimate the likelihood of metabolic pharmacokinetic interactions. Clin Pharmacokinet 1997; 32(3): 210–58
Mihaly GW, Moore G, Thomas J, et al. The pharmacokinetics and metabolism of the anilide local anaesthetics in neonates. Eur J Clin Pharmacol 1978; 13(2): 143–52
Orlando R, Piccoli P, De Martin S, et al. Cytochrome P450 1A2 is a major determinant of lidocaine metabolism in vivo: effects of liver function. Clin Pharmacol Ther 2004; 75(1): 80–8
Valko K, Nunhuck S, Bevan C, et al. Fast gradient HPLC method to determine compounds binding to human serum albumin: relationships with octanol/water and immobilized artificial membrane lipophilicity. J Pharm Sci 2003; 92(11): 2236–48
Dorne JL, Walton K, Renwick AG. Uncertainty factors for chemical risk assessment: human variability in the pharmacokinetics of CYP1A2 probe substrates. Food Chem Toxicol 2001; 39(7): 681–96
DrugBank. Levetiracetam [online]. Available from URL: http://redpoll.pharmacy.ualberta.ca/drugbank/cgi-bin/getCard.cgi?CARD=APRD01068.txt [Accessed 2007 Dec 1]
Nicolas JM, Collart P, Gerin B, et al. In vitro evaluation of potential drug interactions with levetiracetam, a new antiepileptic agent. Drug Metab Dispos 1999; 27(2): 250–4
Ferrier C, Marty J, Bouffard Y, et al. Alfentanil pharmacokinetics in patients with cirrhosis. Anesthesiology 1985; 62(4): 480–4
Kim WR, Poterucha JJ, Wiesner RH, et al. The relative role of the Child-Pugh classification and the Mayo natural history model in the assessment of survival in patients with primary sclerosing cholangitis. Hepatology 1999; 29(6): 1643–8
Orlando R, Piccoli P, De MS, et al. Effect of the CYP3A4 inhibitor erythromycin on the pharmacokinetics of lignocaine and its pharmacologically active metabolites in subjects with normal and impaired liver function. Br J Clin Pharmacol 2003; 55(1): 86–93
Shi BM, Wang XY, Mu QL, et al. Value of portal hemodynamics and hypersplenism in cirrhosis staging. World J Gastroenterol 2005; 11(5): 708–11
Denys A, Menu Y. Portal and splanchnic blood flow measurements in vivo: US Doppler or MR angiography? J Hepatol 1997; 26(2): 437–8
Lautt WW. Mechanism and role of intrinsic regulation of hepatic arterial blood flow: hepatic arterial buffer response. Am J Physiol 1985; 249 (5 Pt 1): G549–56
Braillon A, Cales P, Valla D, et al. Influence of the degree of liver failure on systemic and splanchnic haemodynamics and on response to propranolol in patients with cirrhosis. Gut 1986; 27(10): 1204–9
Carrella M, Hunter JO, Fazio S, et al. Capillary blood flow to the skin of forearm in cirrhosis. Angiology 1992; 43(12): 969–74
Acknowledgements
No specific funding was received for this study. The authors are employed by Bayer Technology Services GmbH (Leverkusen, Germany).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Edginton, A.N., Willmann, S. Physiology-Based Simulations of a Pathological Condition. Clin Pharmacokinet 47, 743–752 (2008). https://doi.org/10.2165/00003088-200847110-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-200847110-00005